Chapt pter er 14: Coval valent ent Bondin ding: g: Orbita tals ls
Chapter 14: Phenomena
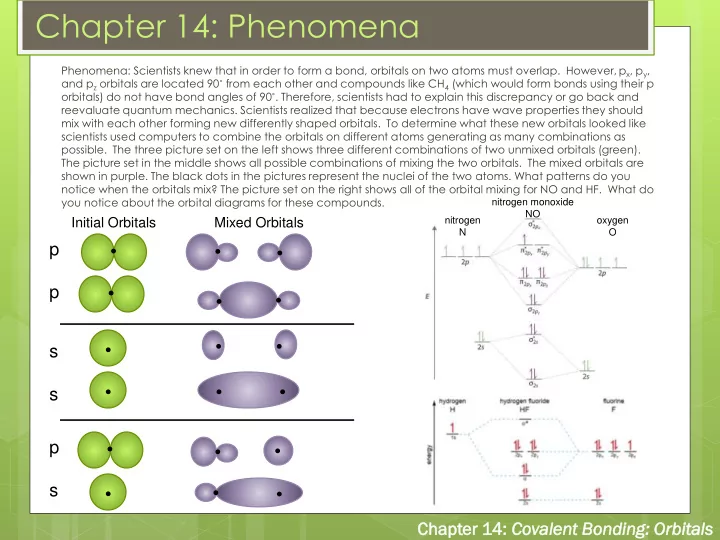
Phenomena: Scientists knew that in order to form a bond, orbitals on two atoms must overlap. However, px, py, and pz orbitals are located 90˚ from each other and compounds like CH4 (which would form bonds using their p
- rbitals) do not have bond angles of 90˚. Therefore, scientists had to explain this discrepancy or go back and
reevaluate quantum mechanics. Scientists realized that because electrons have wave properties they should mix with each other forming new differently shaped orbitals. To determine what these new orbitals looked like scientists used computers to combine the orbitals on different atoms generating as many combinations as
- possible. The three picture set on the left shows three different combinations of two unmixed orbitals (green).
The picture set in the middle shows all possible combinations of mixing the two orbitals. The mixed orbitals are shown in purple. The black dots in the pictures represent the nuclei of the two atoms. What patterns do you notice when the orbitals mix? The picture set on the right shows all of the orbital mixing for NO and HF. What do you notice about the orbital diagrams for these compounds.
Initial Orbitals Mixed Orbitals
p p s s
nitrogen N
- xygen
O nitrogen monoxide NO