Chapt pter er 13 13: Bondi nding: g: General eral Concep epts ts
Chapter 13: Phenomena
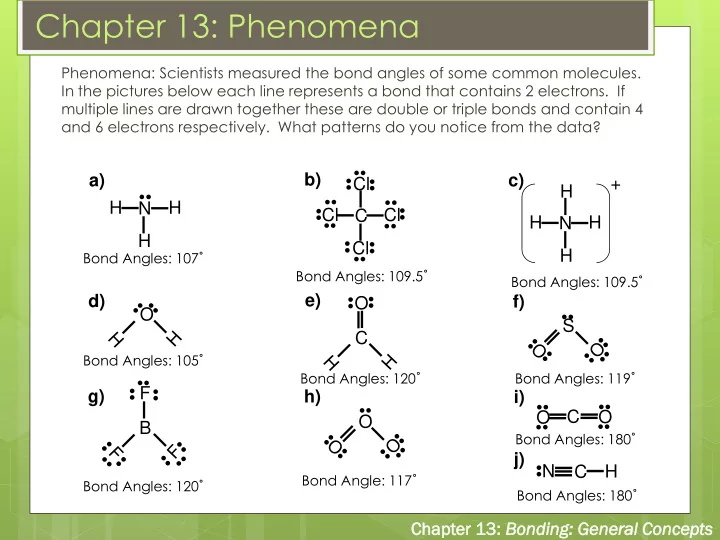
Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together these are double or triple bonds and contain 4 and 6 electrons respectively. What patterns do you notice from the data?
H H N H
H H N H H + O C O
O
C N
H
Cl Cl C Cl Cl
B F
Bond Angles: 107˚ Bond Angles: 109.5˚ Bond Angles: 109.5˚ Bond Angles: 120˚
C O
Bond Angles: 105˚ Bond Angles: 119˚ Bond Angles: 120˚ Bond Angle: 117˚ Bond Angles: 180˚ Bond Angles: 180˚
a) b) c) d) g) e) h) f) i) j) S
O