Chapt pter er 11: Electr troc
- chem
emis istr try
Chapter 11: Phenomena
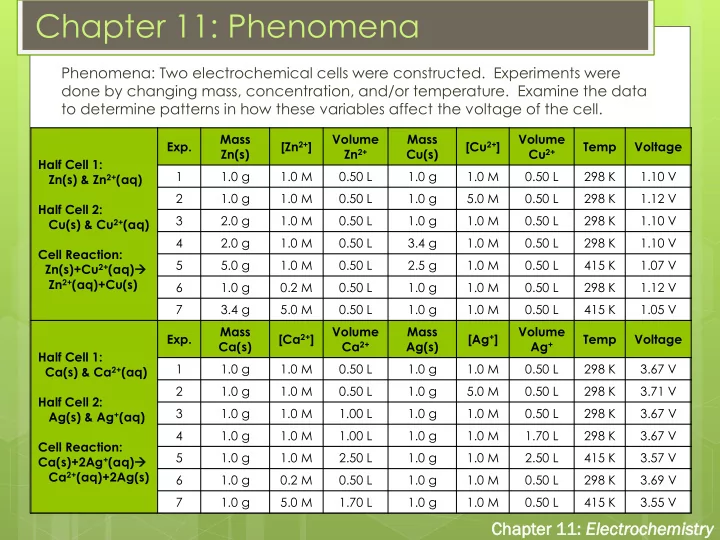
Phenomena: Two electrochemical cells were constructed. Experiments were done by changing mass, concentration, and/or temperature. Examine the data to determine patterns in how these variables affect the voltage of the cell.
Half Cell 1: Zn(s) & Zn2+(aq) Half Cell 2: Cu(s) & Cu2+(aq) Cell Reaction: Zn(s)+Cu2+(aq) Zn2+(aq)+Cu(s) Exp. Mass Zn(s) [Zn2+] Volume Zn2+ Mass Cu(s) [Cu2+] Volume Cu2+ Temp Voltage 1 1.0 g 1.0 M 0.50 L 1.0 g 1.0 M 0.50 L 298 K 1.10 V 2 1.0 g 1.0 M 0.50 L 1.0 g 5.0 M 0.50 L 298 K 1.12 V 3 2.0 g 1.0 M 0.50 L 1.0 g 1.0 M 0.50 L 298 K 1.10 V 4 2.0 g 1.0 M 0.50 L 3.4 g 1.0 M 0.50 L 298 K 1.10 V 5 5.0 g 1.0 M 0.50 L 2.5 g 1.0 M 0.50 L 415 K 1.07 V 6 1.0 g 0.2 M 0.50 L 1.0 g 1.0 M 0.50 L 298 K 1.12 V 7 3.4 g 5.0 M 0.50 L 1.0 g 1.0 M 0.50 L 415 K 1.05 V Half Cell 1: Ca(s) & Ca2+(aq) Half Cell 2: Ag(s) & Ag+(aq) Cell Reaction: Ca(s)+2Ag+(aq) Ca2+(aq)+2Ag(s) Exp. Mass Ca(s) [Ca2+] Volume Ca2+ Mass Ag(s) [Ag+] Volume Ag+ Temp Voltage 1 1.0 g 1.0 M 0.50 L 1.0 g 1.0 M 0.50 L 298 K 3.67 V 2 1.0 g 1.0 M 0.50 L 1.0 g 5.0 M 0.50 L 298 K 3.71 V 3 1.0 g 1.0 M 1.00 L 1.0 g 1.0 M 0.50 L 298 K 3.67 V 4 1.0 g 1.0 M 1.00 L 1.0 g 1.0 M 1.70 L 298 K 3.67 V 5 1.0 g 1.0 M 2.50 L 1.0 g 1.0 M 2.50 L 415 K 3.57 V 6 1.0 g 0.2 M 0.50 L 1.0 g 1.0 M 0.50 L 298 K 3.69 V 7 1.0 g 5.0 M 1.70 L 1.0 g 1.0 M 0.50 L 415 K 3.55 V