Cha hapt pter er 7: Acid ids s and Ba Bases ses
Chapter 7: Phenomena
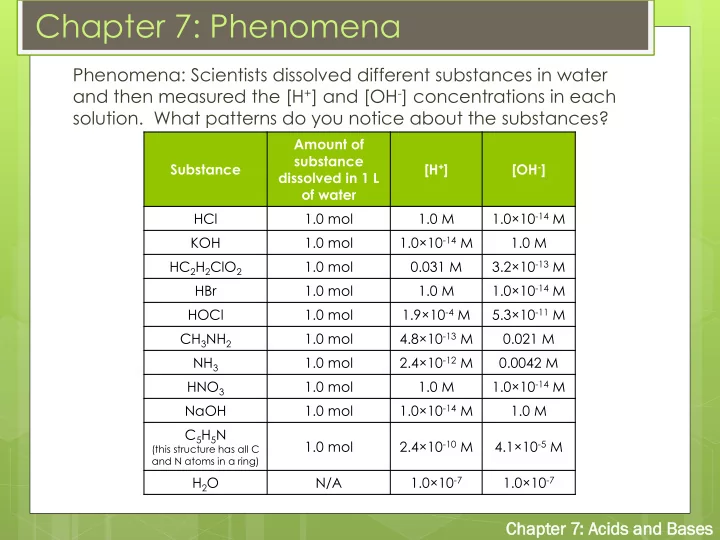
Phenomena: Scientists dissolved different substances in water and then measured the [H+] and [OH-] concentrations in each
- solution. What patterns do you notice about the substances?
Substance Amount of substance dissolved in 1 L
- f water
[H+] [OH-] HCl 1.0 mol 1.0 M 1.0×10-14 M KOH 1.0 mol 1.0×10-14 M 1.0 M HC2H2ClO2 1.0 mol 0.031 M 3.2×10-13 M HBr 1.0 mol 1.0 M 1.0×10-14 M HOCl 1.0 mol 1.9×10-4 M 5.3×10-11 M CH3NH2 1.0 mol 4.8×10-13 M 0.021 M NH3 1.0 mol 2.4×10-12 M 0.0042 M HNO3 1.0 mol 1.0 M 1.0×10-14 M NaOH 1.0 mol 1.0×10-14 M 1.0 M C5H5N
(this structure has all C and N atoms in a ring)
1.0 mol 2.4×10-10 M 4.1×10-5 M H2O N/A 1.0×10-7 1.0×10-7