Chapt pter er 9: Energy, Enthal halpy, and d Therm ermoc
- chem
emis istr try
Chapter 9: Phenomena
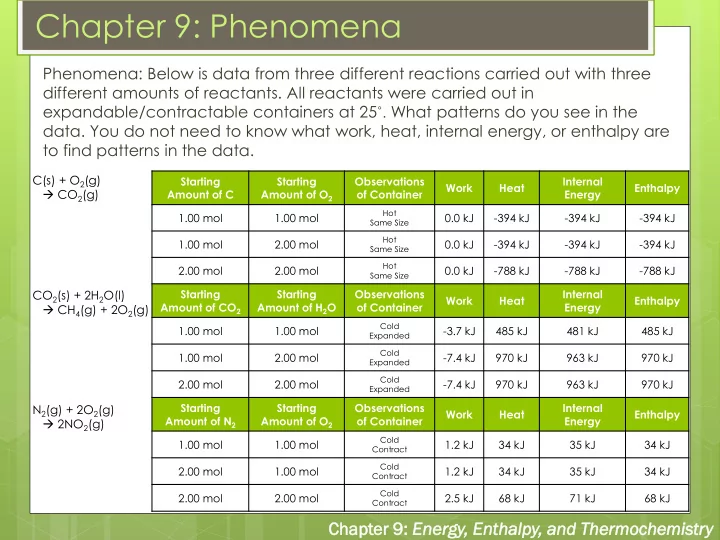
Phenomena: Below is data from three different reactions carried out with three different amounts of reactants. All reactants were carried out in expandable/contractable containers at 25˚. What patterns do you see in the
- data. You do not need to know what work, heat, internal energy, or enthalpy are
to find patterns in the data.
C(s) + O2(g) CO2(g) CO2(s) + 2H2O(l) CH4(g) + 2O2(g)
Starting Amount of C Starting Amount of O2 Observations
- f Container
Work Heat Internal Energy Enthalpy 1.00 mol 1.00 mol
Hot Same Size
0.0 kJ
- 394 kJ
- 394 kJ
- 394 kJ
1.00 mol 2.00 mol
Hot Same Size
0.0 kJ
- 394 kJ
- 394 kJ
- 394 kJ
2.00 mol 2.00 mol
Hot Same Size
0.0 kJ
- 788 kJ
- 788 kJ
- 788 kJ
Starting Amount of CO2 Starting Amount of H2O Observations
- f Container
Work Heat Internal Energy Enthalpy 1.00 mol 1.00 mol
Cold Expanded
- 3.7 kJ
485 kJ 481 kJ 485 kJ 1.00 mol 2.00 mol
Cold Expanded
- 7.4 kJ
970 kJ 963 kJ 970 kJ 2.00 mol 2.00 mol
Cold Expanded
- 7.4 kJ
970 kJ 963 kJ 970 kJ Starting Amount of N2 Starting Amount of O2 Observations
- f Container
Work Heat Internal Energy Enthalpy 1.00 mol 1.00 mol
Cold Contract
1.2 kJ 34 kJ 35 kJ 34 kJ 2.00 mol 1.00 mol
Cold Contract
1.2 kJ 34 kJ 35 kJ 34 kJ 2.00 mol 2.00 mol
Cold Contract
2.5 kJ 68 kJ 71 kJ 68 kJ
N2(g) + 2O2(g) 2NO2(g)