SLIDE 1 YSU YSU
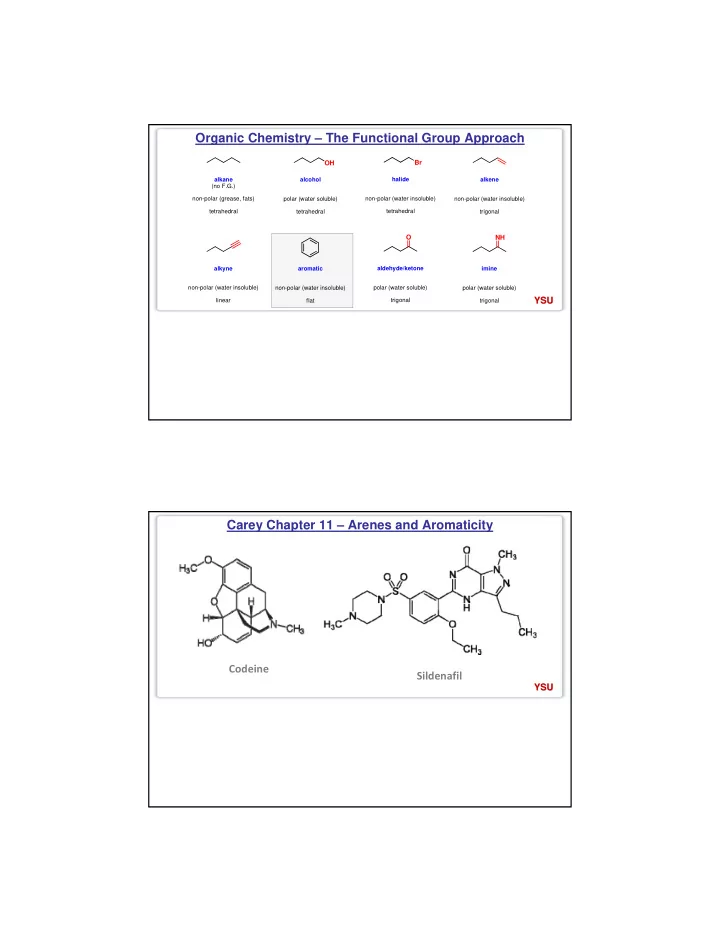
Organic Chemistry – The Functional Group Approach
alkane (no F.G.) non-polar (grease, fats) tetrahedral
OH
alcohol polar (water soluble) tetrahedral
Br
halide non-polar (water insoluble) tetrahedral alkene non-polar (water insoluble) trigonal alkyne non-polar (water insoluble) linear aromatic non-polar (water insoluble) flat aldehyde/ketone polar (water soluble) trigonal imine polar (water soluble) trigonal
O NH
YSU YSU
Carey Chapter 11 – Arenes and Aromaticity
Codeine Sildenafil

SLIDE 2
YSU YSU
11.1 – Increasing Unsaturation in 6-Membered Rings
YSU YSU
11.2 – Evidence of Structure for Benzene all C‐C bonds are the same length, all H’s are equivalent
Kekule (1866) – two rapidly interconverting isomers?

SLIDE 3
YSU YSU
11.2 – Evidence of Structure for Benzene all C‐C bonds are the same length, all H’s are equivalent
Robinson (1920) ‐ the two Kekule forms are resonance contributors
YSU YSU
11.2 – Evidence of Structure for Benzene all C‐C bonds are the same length, all H’s are equivalent
Robinson depiction : “Aromatic Sextet”

SLIDE 4 YSU YSU
11.4 Resonance energy of benzene as estimated from heats
Figure 11.2
Benzene is a lot more stable than “cyclohexatriene” YSU YSU
11.5 – The s bonds (a), the delocalized p system (b), and the electrostatic potential map (c) of benzene
i.e. each carbon experiences the same electron density, the six pi electrons are delocalized over the entire molecule
Figure 11.3

SLIDE 5 YSU YSU
11.6 – The molecular orbitals of benzene arranged in order
Figure 11.4
YSU YSU
Figure 11.4

SLIDE 6
YSU YSU
11.7 – Nomenclature of Substituted Benzenes
Many have common names, however IUPAC systematic names often easier to work out
YSU YSU
11.7 – Nomenclature of Disubstituted Benzenes
Br CH3 CH3 NO2 F NH2 Br CH3 CH3 CH2H3 F Br CH3 Can use numbering or o, m, p nomenclature systems

SLIDE 7
YSU YSU
Not Covering
11.8 11.9 11.11
11.12 – Free-Radical Halogenation of Alkylbenzenes
YSU YSU
C H H H C H H
+ H
C H H H
+ H
C H H C H H H
+ H
C H H
H = 91 kcal/mol H = 88 kcal/mol H = 85 kcal/mol

SLIDE 8
YSU YSU
11.12 – Free-Radical Halogenation of Alkylbenzenes
Figure 11.9 C H H H C H H
Br Br2
CCl4, 80 oC (+ HBr) 71% yield
YSU YSU
11.13 – Oxidation of Alkylbenzenes

SLIDE 9
YSU YSU
11.14 – Nucleophilic Substitution in Benzylic Halides
SN2 applies with good nucleophiles on 1o and 2o carbons SN1 applies with weak nucleophiles – good carbocation E2 competes with more basic nucleophiles on 2o and 3o
YSU YSU
11.15 – Preparation of Alkenylbenzenes

SLIDE 10
YSU YSU
11.16 – Addition to Alkenylbenzenes
YSU YSU
Not Covering 11.17 11.18

SLIDE 11
YSU YSU
11.19 – Hückel’s Rule
http://redandr.ca/vm3/Heme.jpg YSU YSU
11.19 – Hückel’s Rule
N S O
Aromatic = 4n+2 electrons and flat system

SLIDE 12
YSU YSU
11.19 – Hückel’s Rule
Figure 11.12
YSU YSU
Not Covering 11.20

SLIDE 13
YSU YSU
11.21 – Aromatic Ions
Cation is relatively easy to form: 4n + 2 = 6 system capable of being flat
Figure 11.13
YSU YSU
11.21 – Aromatic Ions
pKa of acid is ~16 since anion is aromatic: 4n + 2 = 6 system capable of being flat
Figure 11.14

SLIDE 14
YSU YSU
11.23 – Heterocyclic Aromatic Compounds – Hückel’s Rule
Figure 11.15
N N H pyridine pyrrole
.. ..