SLIDE 1
DEHYDROGENATIVE TRANSFORMATION OF ALCOHOLS Ar Ar Ar Ar P P - - PowerPoint PPT Presentation
DEHYDROGENATIVE TRANSFORMATION OF ALCOHOLS Ar Ar Ar Ar P P - - PowerPoint PPT Presentation
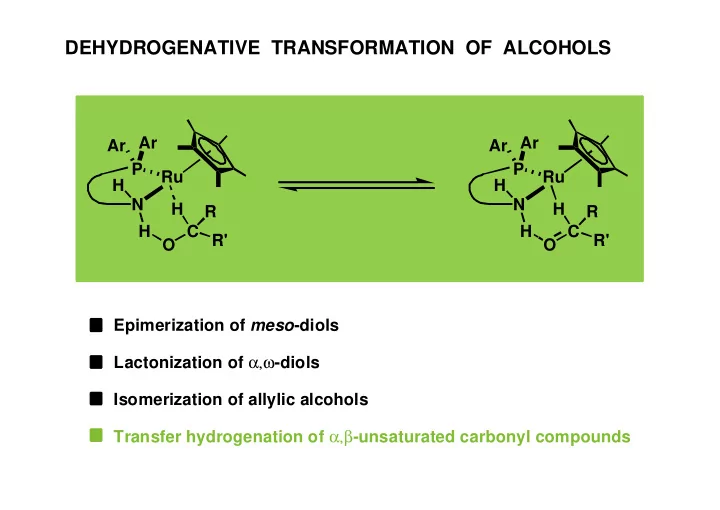
DEHYDROGENATIVE TRANSFORMATION OF ALCOHOLS Ar Ar Ar Ar P P Ru Ru H H N N H H R R H C R' H C R' O O Epimerization of meso -diols Lactonization of , -diols Isomerization of allylic alcohols Transfer hydrogenation of
SLIDE 2
SLIDE 3
ENONE EXCHANGE REACTION
Ph OH O Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu C6D6 0.5 M, 30 °C, 1 h >99% conv. alcohol:enone:Ru:KOt-Bu = 100:100:1:1 Ph O O O Ph 24 % 76 % + O + Intramolecular Intermolecular
SLIDE 4
R1 O Ph Ph O Ph Ph O Ph R1 O Ph
TRANSFER HYDROGENATION OF ACTIVATED OLEFIN WITH ALCOHOL
RuCl2(PPh3)3, 0.25 mol% PhCH(OH)CH3, 180 °C, 1 h Sasson et al. J. Org. Chem., 1975, 40, 1887 92% [Ir(cod)Cl]2/dppp/ Cs2CO3, 2 mol% toluene Sakaguchi et al. J. Org. Chem., 2001, 66, 4710 [RuH((S)-binap)2]PF6, 2 mol% EtOH or 2-propanol, reflux, 24 h >99% Saburi et al.Tetrahedron Lett., 1992, 33, 5783 R1 = CH2COOH R1 = NHCOCH3 R2 = H or CH3 Edwards et al. Angew. Chem. Int. Ed., 2002, 41, 4740 + + + HO O R R1 HO O R R1 OH R2 R3 O R2 R3 R1 = CH3, R2 = CH3, R3 = CH3 80 °C, 4 h 93% 92% + + OH R2 O R2 OH Ph O Ph + R1 = OBn, R2 = Ph, R3 = H 150 °C, 72 h
SLIDE 5
TRANSFER HYDROGENATION OF ENONEs USING 2-PROPANOL
R O R3 O O R1 R2 R O R3 R1 R2 Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu toluene 30 °C, 1 h O Ph O 38% endo:exo = >99:<1 >99% yield >99% 47% 52% + OH + O enone:alcohol:Ru:KOt-Bu = 100:100:1:1 [enone] = 0.5 M O
SLIDE 6
R1 ROCO R3 Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu toluene 0.5 M, 30 °C, 1 h R1 ROCO R3 EtOCO EtOCO n-BuOCO O O O O 54 % 13 % R2 R2 + + OH O CH3OCO CH3OCO EtOCO BnOCO EtOCO CO2Et EtOCO >99% yield >99% >99% >99% 54% 4% 4% 41%
TRANSFER HYDROGENATION OF α,β−UNSATURATED ESTERs WITH ALCOHOL
2-propanol large excess 10 equiv. ester:Ru:KOt-Bu = 100:1:1 0%
SLIDE 7
ISOTOPE LABELING EXPERIMENT
BnOCO + CD3 CD3 OD Cp*RuCl[Ph2P(CH2)2NH2] KOt-Bu toluene 0.5 M, 30 °C, 2 h, BnOCO + CD3 CD3 O α β 87% conv. α position; 0.86 D β position; 1.00 D D D D loss of deuterium ! determined by 1H & 2H NMR. C6D6 as an internal standard (2H NMR) ester:alcohol:Ru:KOt-Bu = 100:100:1:1
SLIDE 8
DISTRIBUTION OF DEUTERIUM
BnOCO BnOCO BnOCO + 0.86 D 1.00 D cis; 0.31 D trans; 0.31 D [Ru] BnOCO H H [Ru] β-hydride elimination [Ru] BnOCO H D D H H BnOCO H D
SLIDE 9