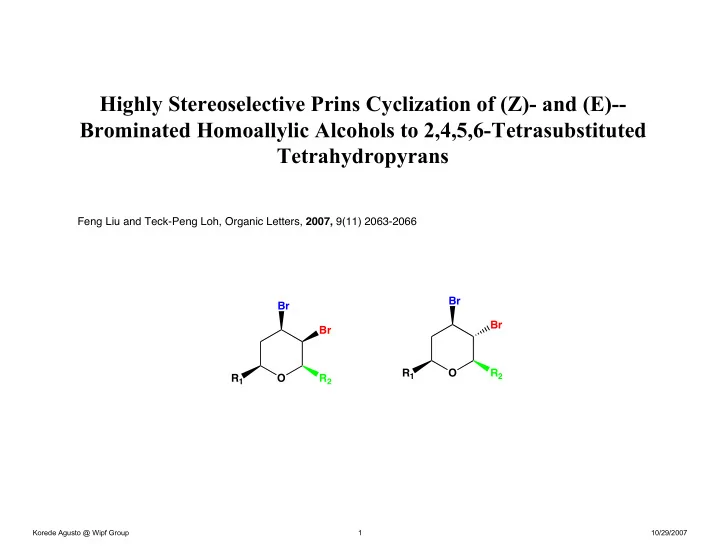
Highly Stereoselective Prins Cyclization of (Z)- and (E)-- Brominated Homoallylic Alcohols to 2,4,5,6-Tetrasubstituted Tetrahydropyrans
Feng Liu and Teck-Peng Loh, Organic Letters, 2007, 9(11) 2063-2066 O O R1 R1 Br Br R2 Br R2 Br
Korede Agusto @ Wipf Group 1 10/29/2007