SLIDE 1
Cp*Ru(PN) CATALYSTS WITH METAL/NH BIFUNCTION R 1 R 1 Ru Ru H H - - PowerPoint PPT Presentation
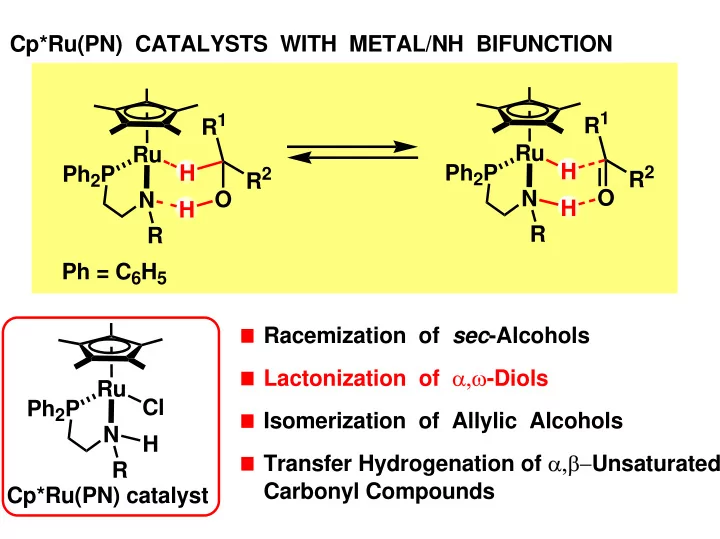
Cp*Ru(PN) CATALYSTS WITH METAL/NH BIFUNCTION R 1 R 1 Ru Ru H H - - PowerPoint PPT Presentation
Cp*Ru(PN) CATALYSTS WITH METAL/NH BIFUNCTION R 1 R 1 Ru Ru H H Ph 2 P Ph 2 P R 2 R 2 O O N N H H R R Ph = C 6 H 5 Racemization of sec -Alcohols Lactonization of , -Diols Ru Cl Ph 2 P Isomerization of Allylic Alcohols N H
SLIDE 2
SLIDE 3
OH OH + O 2 O O OH 2 + Ru cat KOt-C4H9 acetone
Regioselective lactonization of 2-substituted 1,4-butanediols R R
O +
R
O
1 2 4 4 1 2 4 1 2
LACTONIZATION OF UNSYMMETRICAL 1,4-DIOL
diol:Ru cat:KOt-C4H9 = 100:1:1, [diol] = 0.5 M OH OH + O 2 Cp*RuCl(PN) KOt-C4H9 PN = Ph2P(CH2)2NH2 30 °C, 1 h O + OH 2 R2 R1 R1 R2 O acetone The 79th Annual Meeting of Chemical Society of Japan. 2001, 1H326 R1 R2 yield, % CH3 CH3 CH3 CH3 H D (96%) >99 >99 (96% atom D) >99
THIS WORK
SLIDE 4
REPORTED LACTONIZATION OF UNSYMMETRICAL DIOLS
OH OH Ph O CH3 R R' + Ishii, Y.; Osakada, K.; Ikariya, T.; Saburi, M.; Yoshikawa, S. J. Org. Chem. 1986, 51, 2034 R R' R R' Ph O CH3 O O O Ru cat toluene + + O R = CH3; R' = CH3 A B R = i-(C3H7); R' = H R = Ph; R' = H 20 °C, 10 h yield, % A:B 100 100 90 99.6:0.4 98:2 97:3 OH OH Ph Ph Ir cat acetone rt, 20 h diol:Ir cat = 200:1 Ph Ph Ph Ph O O O + O HN O Ph Ph Ir cat 98% yield RuH2(PPh3)4 Ru cat A B A:B = >99:1 Suzuki, T.; Morita, K.; Tsutida, M.; Hiroi, K. Org. Lett. 2002, 4, 2361 diol:Ru cat = 25:1 2 2 Ir
SLIDE 5
78:22
LIGAND EFFECT:Cp STRUCTURE
OH OH Ph Ru cat KOt-C4H9 acetone 30 °C, 1 h A B Ph O O Ph O O + diol:Ru cat:KOt-C4H9 = 100:1:1, [diol] = 0.5 M yield, % A:B* >99 >99 56:44 Ru NCCH3 Ph2 P N H2 PF6 * Determined by 1H NMR analysis Ru NCCH3 Ph2 P N H2 OTf Ph = C6H5 Ru cat
SLIDE 6
LIGAND EFFECT: N- & P- & N-α-SUBSTITUENTS
A:B Ru cat 78:22 82:18 83:17 P N 81:19 76:24 78:22 81:19 (4-Tol)2 (2,6-Xy)2 A:B P N Ru NCCH3 P N OTf NH2 Ph2P NHCH3 Ph2P NHCH2Ph Ph2P NH2 Ph2P Ph NH2 Ph2P CH2Ph NH2 P CH2Ph NH2 P CH2Ph 4-Tol 2,6-Xy
SLIDE 7
LIGAND EFFECT: BACKBONE
OH OH Ph Ru cat KOt-C4H9 acetone 30 °C, 1 h A B Ph O O Ph O O + diol:Ru cat:KOt-C4H9 = 100:1:1, [diol] = 0.5 M A:B P N 92:8
P NH2 Ph2
82:18
P NH2 Ph2
92:8 92:8
P NCH3 Ph2 P NCH2Ph Ph2 H H
47 20 yield, % >99 >99 Ru cat Ru NCCH3 P N OTf
SLIDE 8
EFFECT OF ARYLMETHYL SUBSTITUENTS IN 1,4-DIOL
A:B 92:8 92:8 92:8 OH OH Ar Ru cat KOt-C4H9 acetone 30 °C, 1 h O O O Ar Ar O + A B O O CH3O NCCH3 Ph2 P N H2 OTf Ru cat diol:Ru cat:KOt-C4H9 = 100:1:1, [diol] = 0.5 M >99% yield PhCH2O 92:8 Ar Ru
SLIDE 9
(−)-Hinokinin
BIOLOGICALLY IMPORTANT LIGNANS
O O O O O O O O Rn O O HO HO (−)-Enterolactone O O O O (−)-Podophyllotoxin CH3O OCH3 OCH3 OH O O O O CH3O OCH3 OCH3 O O O O OH HO CH3 (+)-Isostegane O OCH3 O O O O O CH3 CH3 Etoposide
SLIDE 10
SYNTHESIS OF JUSTICIDIN E
Ru cat KOt-C4H9 acetone 30 °C, 15 h diol:Ru cat:KOt-C4H9=50:1:1 OH OH O O O O O O O O O O O O O O O O 94% 0% Justicidin E Taiwanin C + NCCH3 Ph2 P N H2 OTf Ru cat Ru
SLIDE 11