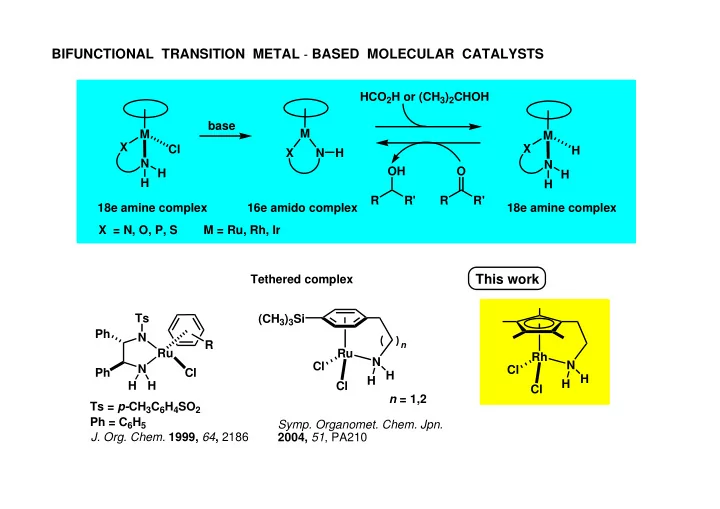
SLIDE 1 M X N Cl M X N X = N, O, P, S M X N H H H H base 18e amine complex 16e amido complex 18e amine complex Ts = p-CH3C6H4SO2
BIFUNCTIONAL TRANSITION METAL-BASED MOLECULAR CATALYSTS
Ph = C6H5
This work
n = 1,2 Tethered complex H H Ru N H H Cl Cl (CH3)3Si N Ru N Ph Ph H H Ts
n
R Cl Rh N H H Cl Cl
- Symp. Organomet. Chem. Jpn.
2004, 51, PA210 R R' O R R' OH HCO2H or (CH3)2CHOH M = Ru, Rh, Ir
- J. Org. Chem. 1999, 64, 2186

SLIDE 2
Isomerization of the cyclic amine HCl in ether (1.5 equiv) CH3OH 50 °C, 2 h 78% yield NH2 NH3Cl NH3Cl NH3Cl + +
SYNTHESIS OF Rh DIMER BEARING AMINOALKYL SIDE CHAIN
Rh Cl Rh Cl ClH3N NH3Cl Cl Cl NH3Cl + CH3OH reflux, 66 h RhCl3・3H2O Orange powder 83% yield 1 : 3
1H NMR (DMSO-d6)
δ7.87 (br, 3H, NH3) 2.92 (br, 2H, CH2NH3) 2.41 (t, 2H, J = 8.2 Hz, CH2CH2NH3) 1.71 (s, 6H, CH3) 1.63 (s, 6H, CH3)
13C NMR (DMSO-d6)
δ102.1, 98.6, 93.3 (each d,C5Me4) 36.0 (CH2NH3) 21.6 (CH2CH2NH3) 8.8, 8.5 (each CH3) Leusen, D.; Beetstra, D. J.; Hessen, B.; Teuben, J. H. Organometallics 2000, 19, 4084

SLIDE 3
SYNTHESIS OF MONONUCLEAR TETHERED Rh COMPLEX
1H NMR (CDCl3)
δ3.90 (m, 2H, CH2NH2) 3.59 (br, 2H, NH2) 2.41 (t, 2H, 3J = 6.7 Hz, CH2CH2NH2) 1.90 (s, 6H, CH3) 1.69 (s, 6H, CH3) KOtC4H9 CH2Cl2 rt, 9 h 25% yield
13C NMR (CDCl3)
δ109.1 (d,C5Me4) 97.5 (d, C5Me4) 85.8 (d, C5Me4) 58.0 (s, CH2NH2) 25.4 (s, CH2CH2NH2) 9.1 (s, CH3) 8.9 (s,CH3) Rh Cl Rh Cl ClH3N NH3Cl Cl Cl Spectra data of new complex Rh N H H Cl Cl

SLIDE 4
Rh Cl Cl
2
ClH3N Rh Cl Ph3P Cl 78% yield NH3Cl 1) KOtC4H9 2) AgSbF6 33% yield Rh N H H Cl Ph3P SbF6 PPh3 (after recrystallization)
SYNTHESIS OF CATIONIC TETHERED COMPLEX AND ITS SPECTRAL DATA
1H NMR (CD2Cl2)
δ7.43-7.58 (m, 15H, C6H5) 3.94 (m, 1H) 3.77 (m, 2H) 2.67 (m, 2H) 2.20-2.28 (m, 1H) 2.13 (d, 3H, 4JHP = 7.6 Hz, CH3) 1.69 (s, 3H, CH3) 1.21 (s, 3H, CH3) 0.97 (dd, 3H, J = 0.7, 5.4 Hz, CH3)
31P NMR (CD2Cl2)
δ33.1 (d, JPRh = 141.0 Hz)
1H NMR (DMSO-d6)
δ7.85 (br, 3H, NH3) 7.71 (m, 6H, C6H5) 7.45 (m, 9H, C6H5) 2.86 (br, 2H, CH2NH3) 2.29 (m, 2H, CH2CH2NH3) 1.38 (d, 6H, 4JHP = 3.2 Hz, CH3) 1.07 (d, 6H, 4JHP = 3.4 Hz, CH3) δ30.0 (d, JPRh = 144.6 Hz)
31P NMR (DMSO-d6)
CH3CH2OH reflux, 7 h THF rt, 1) 0.5 h 2) 2 h

SLIDE 5
STRUCTURE OF TETHERED Rh COMPLEX
1H NMR (CDCl3)
δ7.59 (m, 2H, C6H4) 6.78 (m, 2H, C6H4) 4.14 (br, 4H, CH2NH2) 2.42 (t, 2H, J = 6.3 Hz, CH2CH2NH2) 1.86 (s, 6H, CH3) 1.37 (s, 6H, CH3)
19F NMR (CDCl3)
δ& 75.8 (SO2CF3) Tf = SO2CF3 P21/a (#14) R1 = 0.033 wR2 = 0.120 Rh Cl Rh Cl ClH3N NH3Cl Cl Cl + KOtC4H9 (3 equiv/Rh) NHTf NHTf 1 : 2 84% yield Rh N H H NTf TfN THF reflux, 4 h

SLIDE 6
O OH Rh cat Rh cat O NH2 OH + HCOONH4 Rh cat ketone : Rh = 100 : 1 < 1% conv + HCO2H・N(C2H5)3 ketone : Rh = 100 : 1 + CH3OH 70 °C
CATAYLTIC ACTIVITY OF TETHERED Rh COMPLEX
Reductive amination of acetophenone Rh cat > 99 : < 1 38 TOF (h-1) Reduction of acetophenone 5 equiv 5 equiv Rh N H H Cl Cl Rh N H H Cl Cl CH3OH 70 °C, 6 h

SLIDE 7 CATALYTIC LEUCKART-WALLACH TYPE REDUCTIVE AMINATION OF KETONES
Rh Cl Cl
2
Rh cat O NH HCOONH4 Possible mechanism NH2 H Kitamura, M.; Lee, D.; Hayashi, S.; Tanaka, S.; Yoshimura, M.
- J. Org. Chem. 2002, 67, 8685
O NH2 OH + HCOONH4 Rh cat ketone : Rh = 100 : 1 + CH3OH 70 °C, 2 h 98 : 2 (98% conv) 5 equiv Rh NH3 H Cl

SLIDE 8
SUMMARY
Synthesis of tethered Rh complex with primary amine Rh Cl Rh Cl ClH3N NH3Cl Cl Cl CH2Cl2 rt, 9 h KOtC4H9 (1 equiv/Rh) Synthesis of analogous tethered Rh complexes Rh N H H Cl Ph3P Rh N H H NTf TfN SbF6 ・Active catalyst for reductive amination Rh N H H Cl Cl Rh N H H Cl Cl