SLIDE 1
TRANSFER HYDROGENATION WITH BIFUNCTIONAL CATALYSTS O OH X X - - PowerPoint PPT Presentation
TRANSFER HYDROGENATION WITH BIFUNCTIONAL CATALYSTS O OH X X - - PowerPoint PPT Presentation
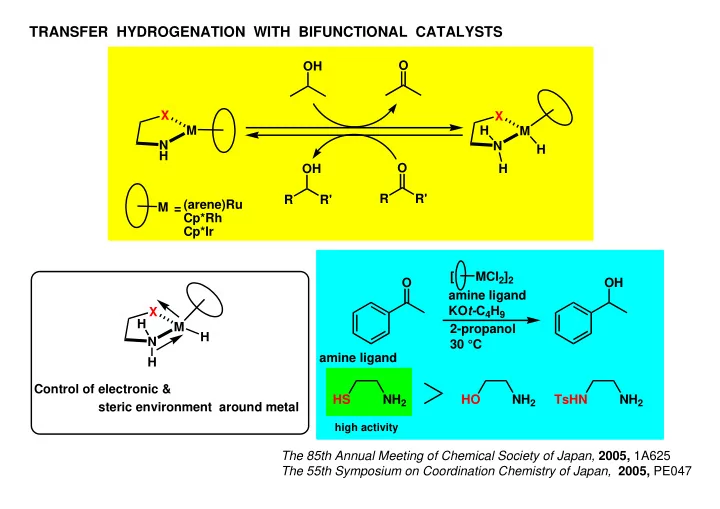
TRANSFER HYDROGENATION WITH BIFUNCTIONAL CATALYSTS O OH X X M H M N N H H O OH H R R' R R' (arene)Ru M = Cp*Rh Cp*Ir [ MCl 2 ] 2 O OH amine ligand KO t- C 4 H 9 X H M 2-propanol H N 30 C amine ligand H
SLIDE 2
SLIDE 3
A POSSIBLE ROUTE FOR GENERATION OF ACTIVE CATALYST
base 3 equiv 16e 18e Ru S NH S Ru Ru S NH HN S Ru Ru S H2N NH3 Cl Cl2 OH OH O O base 2 equiv S Ru Ru S H2N NH2 (OTf)2 Ru S NH2 H –HCl –HOTf
SLIDE 4
H2N SH H2N SH R OH OH O O achiral ligand chiral ligand
THIS WORK
S Ru Ru S H2N NH3 Cl Cl2 S Ru Ru S H2N NH3 Cl Cl2 Ph Ph
SLIDE 5
REPORTED SYNTHESIS OF N-α-SUBSTITUTED SN COMPOUNDS
HO R NH2 Br R NH2 HS R NH2 H2N NH2 S
- H. H. Otto, Helv. Chim. Acta, 2004, 87, 90
HO R HN Boc S R HN Boc O
- B. P. Roques, J. Med. Chem., 1992, 35, 1259
Boc = t-C4H9OCO S NH S R
- T. P. Johnston, J. Med. Chem., 1966, 9, 911
CS2 Br R NH2 HS R HN Boc O SH HBr
SLIDE 6
SYNTHESIS OF N-α-SUBSTITUTED SN LIGANDS
HN O O + H2N S KOt-C4H9 t-C4H9OH reflux R R SH H2N S R ClH3N SH R R = i-C3H7 C6H5 25% 95% 50% 73% CH2C6H5 82% 98% 1) Na/NH3 2) HCl/ether R = i-C3H7 C6H5 CH2C6H5 HN O O + H2N SC6H5 KOt-C4H9 R R C6H5SH Ishibashi, H. Synlett, 1997, 915 t-C4H9OH reflux
SLIDE 7
S Ru Ru S H2N NH3 Cl Cl2 Ph Ph HS NH3Cl Ph [RuCl2(hmb)]2 + KOt-C4H9 [RuCl2(hmb)]2:amine:base = 1:2:3 2-propanol r.t. [Cp*MCl2]2 M = Rh, Ir Ph = C6H5 S M M S H2N NH3 Cl Cl2 Ph Ph HS NH3Cl Ph + KOt-C4H9 [Cp*MCl2]2:amine:base = 1:2:3 2-propanol r.t.
1H NMR (δ, CDCl3):
2.14 (s, 18H, hmb) 8.68 (s, 3H, NH3) 6.05 (s, 1H, NH) 1.95 (s, 18H, hmb) 5.63 (s, 1H, NH)
SYNTHESIS OF CHIRAL THIOLATE–BRIDGED DIMER
ESI-MS: 867
quantitative quantitative
SLIDE 8
ASYMMETRIC TRANSFER HYDROGENATION WITH 2-PROPANOL
KOt-C4H9 O OH metal cat metal cat 30 °C [ketone] = 1 M in 2-propanol S M M S H2N NH3 Cl Cl2 Ph Ph + OH O +
S
ketone:M = 1000:1
2 4 6 8 1 6 1 2 1 8 2 4 r e a c t i
- n
t i m e / h y i e l d / % R u ( h m b ) C p * R h C p * I r 2 4 6 8 1 6 1 2 1 8 2 4 r e a c t i
- n
t i m e / h s e l e c t / % e e R u ( h m b ) C p * R h C p * I r
SLIDE 9
Cp*Rh Cp*Ir
TRANSFER HYDROGENATION WITH FORMIC ACID
N(CH2CH3)3 30 °C O OH metal cat ketone:HCOOH:N(CH2CH3)3 = 2:3:5 metal cat : % ee (yield) at 24 h 52 (74) 65 (46) 75 (19) 76 (81) S M M S H2N NH3 Cl Cl2 Ph Ph 58 (19) 74 (22) + HCOOH + % ee (yield) at 1 h CO2
S
ketone:M = 100:1 M Ru(hmb)
SLIDE 10
S M M S H2N NH3 Cl Cl2 Ph Ph + KOt-C4H9 :amine:base = 1:2:3 2-propanol r.t. HN O O H2N S KOt-C4H9 t-C4H9OH R R C6H5CH2SH ClH3N SH R 1) Na/NH3 2) HCl ClH3N SH Ph HCOOH/N(CH2CH3)3 30 °C O OH (S,S)-Ru2(SN)2 24 h ketone:HCOOH:N(CH2CH3)3 = 2:3:5 75% ee
SUMMARY
Cp*Rh Ru(hmb) Cp*Ir ketone:Ru = 100:1
S
R = C6H5 i-C3H7 CH2C6H5 MCl2]2 [ MCl2]2 [ M =
SLIDE 11