SLIDE 1
MOLECULAR CATALYSTS WITH M/NH BIFUNCTIONAL UNITS Nu H Ru Ru X - - PowerPoint PPT Presentation
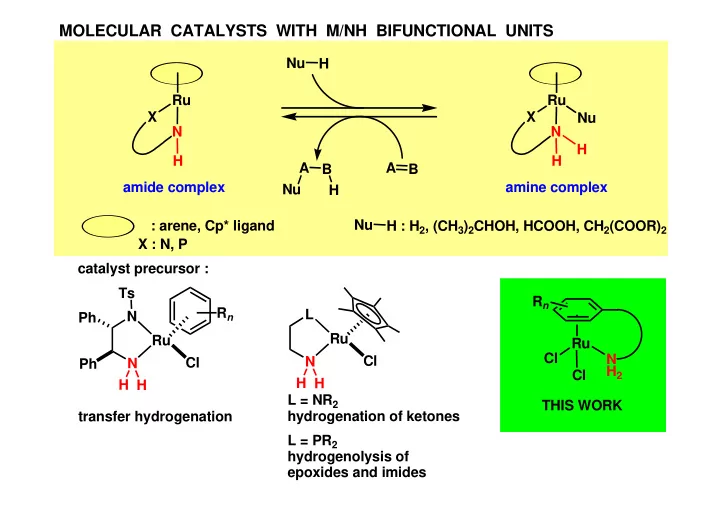
MOLECULAR CATALYSTS WITH M/NH BIFUNCTIONAL UNITS Nu H Ru Ru X - - PowerPoint PPT Presentation
MOLECULAR CATALYSTS WITH M/NH BIFUNCTIONAL UNITS Nu H Ru Ru X X Nu N N H H H A A B B amide complex amine complex Nu H Nu : arene, Cp* ligand H : H 2 , (CH 3 ) 2 CHOH, HCOOH, CH 2 (COOR) 2 X : N, P catalyst precursor : Ts R
SLIDE 2
SLIDE 3
η1−L → η1 : η6 −L−arene
THIS WORK : INTRAMOLECULAR ARENE DISPLACEMENT REACTION
NH2 CO2Et Ru Cl Cl
2
+ η1 −NH2 complex η1:η6−tethered complex ∆ Ru Cl Cl N H2 Ru Cl Cl NH2 EtOCO
SLIDE 4
EFFECT OF SUBSTITUENTS FOR η1: η6-(AMINOARENE)Ru COMPLEXES
chlorobenzene 140 °C Ru Cl Cl N H2 yield, %a H >99 m-CH3 p-CH3 R >99 62 80
- -Si(CH3)3
m-Si(CH3)3 p-Si(CH3)3 88 90 [Ru] = 0.02 M. aIsolated yield. time, h 21 20 2 2 2 18 m,p-(CH3)2 67 17 Ru Cl Cl N H2 EtOCO
R R
SLIDE 5
EFFECT OF STRUCTURE OF AMINOARENES
H
94
m-Si(CH3)3 p-Si(CH3)3
56 16 2 2
NH2 R O Y R yield, %a R p-Si(CH3)3 p-Si(CH3)3 time, h
2 1
Y CO
69 <1 <1
CH2 NH2
NH2
R1 R2 yield, %a R time, h yield, %a R1 time, h R2 CH3 CO2CH3
18 13
H
98 98
CH3 [Ru] = 0.02 M. aIsolated yield.
SLIDE 6
Ru Cl N H2 (CH3)3Si +
1H NMR
ESI-TOF TMS η6-C6H4 Si(CH3)3
Ru Cl Cl N H2 (CH3)3Si
CHCl3
SPECTRAL DATA
SLIDE 7
SYNTHESIS OF CATIONIC ARENE−Ru COMPLEXES
BF4 Ru Ph3P Cl (CH2)2NH3Cl methanol reflux, 20 h 56% yield Ru Cl Cl N H2 Cl methanol rt, 15 h 42% yield this work Ru Ph3P Cl N H2 + + PPh3 + AgBF4 NaOH + NaBF4 Kurosawa, Inorg. Chim. Acta, 2000
SLIDE 8
Ru−C(1) 2.152(9) Ru−C(2) 2.221(9) Ru−C(3) 2.276(8) Ru−C(4) 2.241(7) Ru−C(5) 2.197(6) Ru−C(6) 2.174(7) Ru−Cl 2.404(2) Ru−N 2.157(7) Ru−P 2.360(2) P−Ru−Cl 87.90(7) P−Ru−N 92.0(2) N−Ru−Cl 84.8(2) Selected bond lengths[Å] and angles[°]
STEREOSELECTIVE FORMATION OF PHOSPHINE COMPLEXES
methanol reflux, 18 h PPh3 AgSbF6 Ru Cl Cl N H2 (CH3)3Si Ru Cl PPh3 N H2 SbF6 (CH3)3Si anti 70% yield phosphine complex with ethylene tether single diastereomer (31P NMR) + R1 (I > 2σ) = 0.094 wR2 = 0.175 P-1 (#2) Z = 2
SLIDE 9
Ru−C(1) 2.211(4) Ru−C(2) 2.255(4) Ru−C(3) 2.267(5) Ru−C(4) 2.217(5) Ru−C(5) 2.186(6) Ru−C(6) 2.222(6) Ru−Cl 2.3969(13) Ru−N 2.135(4) Ru−P 2.3541(12) Selected bond lengths[Å] and angles[°] P−Ru−Cl 86.13(4) P−Ru−N 90.93(13) N−Ru−Cl 81.05(13) CH2Cl2 rt, 24 h Ru Cl PPh3 N H2 (CH3)3Si SbF6 Ru Cl Cl N H2 (CH3)3Si anti 52% yield single diastereomer (31P NMR) phosphine complex with propylene tether PPh3 AgSbF6 + R1 (I > 2σ) = 0.053 wR2 = 0.143 P-1 (#2) Z = 4
SLIDE 10
SYNTHESIS OF CATIONIC ISOCYANIDE COMPLEX
Ru Cl Cl N H2 (CH3)3Si Ru Cl CNt-C4H9 N H2 (CH3)3Si + + SbF6 CH2Cl2, rt 2 h, 88% yield IR νN≡C 2201 cm-1 AgSbF6 t-C4H9NC Ru Cl CNt-C4H9 N H2 (CH3)3Si SbF6 IR νN≡C 2197 cm-1 + + CH2Cl2, rt 2 h, 49% yield AgSbF6 t-C4H9NC Ru Cl Cl N H2 (CH3)3Si
SLIDE 11
Ru−Cl 2.3911(13) Ru−N(1) 2.142(3) Ru−C(13) 1.989(4) N(2)−C(13) 1.142(5) C(13)−Ru−Cl 83.34(13) C(13)−Ru−N 86.91(12) N−Ru−Cl 81.62(11) Selected bond lengths[Å] and angles[°] t-C4H9NC AgSbF6 CH2Cl2 rt, 1 h Ru t-C4H9NC Cl N H2 (CH3)3Si SbF6 Ru Cl Cl N H2 (CH3)3Si Ru Cl CNt-C4H9 N H2 (CH3)3Si SbF6 syn 16% yield anti isocyanide complex with propylene tether + IR νN≡C 2193 cm-1 R1 (I > 2σ) = 0.044 wR2 = 0.090 P-1 (#2) Z = 2
SLIDE 12
Si(CH3)3 Ru Cl Cl
2
R1 = n-C6H13 7 9
aDetermined by 1H NMR.
yield, %a
EFFECT OF AMINO-TETHER IN HYDROGEN TRANSFER
O Ru cat t-C4H9OK OH H 2-propanol 30 °C, 1 h ketone:Ru:t-C4H9OK = 10:1:1, [ketone] = 0.1 M Ru Cl Cl N H2 (CH3)3Si Ru Cl Cl N H2 (CH3)3Si 28 >99 yield, %a Ru Cl Cl N H2 (CH3)3Si <1 (without base) Ru Cl Cl NR1 H2 (CH3)3Si
SLIDE 13
aDetermined by 1H NMR.
HYDROGEN TRANSFER USING CATIONIC COMPLEXES
O Ru cat t-C4H9OK OH H 2-propanol 30 °C, 1 h ketone:Ru:t-C4H9OK = 10:1:1, [ketone] = 0.1 M Ru Cl PPh3 N H2 (CH3)3Si >99 10 yield, %a <1 Ru Cl Cl N H2 (CH3)3Si SbF6 Ru Cl CNt-C4H9 N H2 (CH3)3Si SbF6
SLIDE 14
PROPOSED MECHANISM
Ru Cl Cl N H2 base Ru Cl N H Ru Cl L N H2 base Ru Cl N H SbF6 L Ru Cl H N H2 R OH R O H + + – HSbF6 – HCl – L L
SLIDE 15