SLIDE 1
Group VIII & IX Metal Complexes with N,N'- - - PowerPoint PPT Presentation
Group VIII & IX Metal Complexes with N,N'- - - PowerPoint PPT Presentation
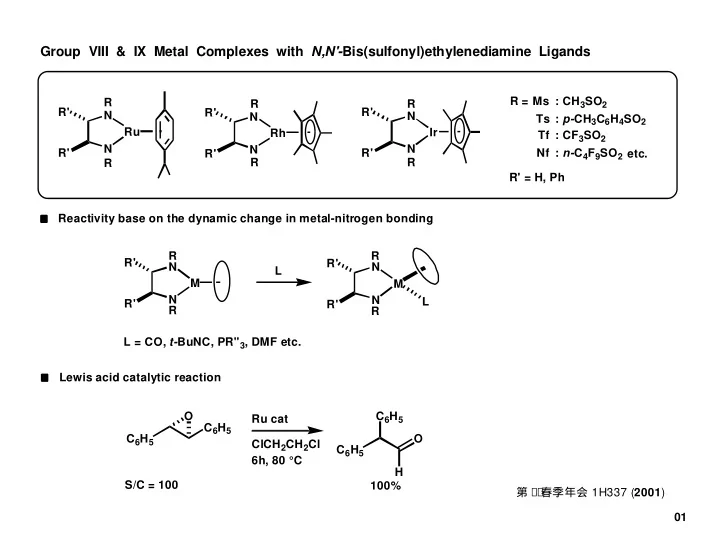
Group VIII & IX Metal Complexes with N,N'- Bis(sulfonyl)ethylenediamine Ligands R = Ms : CH 3 SO 2 R R R R' R' R' N N N Ts : p- CH 3 C 6 H 4 SO 2 Ru Ir Rh Tf : CF 3 SO 2 N N N R' R' Nf : n- C 4 F 9 SO 2 R' etc. R
SLIDE 2
SLIDE 3
Ru N N
Preparation of L4Ru Complexes with N,N'-bis(sulfonyl)ethylenediamine
t-BuNC (20 equiv.) Ru N N CNt-Bu CNt-Bu CNt-Bu CNt-Bu THF, reflux Tf Tf Tf Tf Ru N N Ru N N NCCH3 NCCH3 NCCH3 NCCH3 CH3CN, rt Tf Tf Tf Tf hν (Hg lamp) 1 day 12 h (t-BuNC)4Ru complex (CH3CN)4Ru complex 84% 98% [starting Ru complex] = 1.0 x 10-1 M [starting Ru complex] = 1.0 x 10-2 M C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 03
SLIDE 4
Ru[(S,S)-Tf2dpen](t-BuNC)4 Ru[(S,S)-Tf2dpen](CH3CN)4 side view side view Selected bond lengths(Å) Ru1-C17 Ru1-C22 Ru1-C27 Ru1-C32 Ru1-N1 Ru1-N2 1.936(7) 1.937(7) 2.019(8) 2.023(7) 2.176(5) 2.185(5) Selected bond lengths(Å) Ru1-N2 Ru1-N3 Ru1-N1 2.000(9) 2.026(6) 2.165(8)
168.8(3)° Ru1 N1 N2 C22 C17 C32 C27 Ru1 N2 N2* N1* N1 N3 N3* P1 (#1) R Rw 0.055 0.069 C2 (#5) R Rw 0.050 0.060
04
- ca. 180°
SLIDE 5
Reaction of Ru[(S,S)-Tf2dpen](CH3CN)4 with Various Ligands
Ru N N Tf Tf C6H5 C6H5 NCCH3 NCCH3 NCCH3 NCCH3 CH2Cl2, rt (2.0 equiv.) XylNC 95% Ru N N Tf Tf C6H5 C6H5 NCCH3 NCCH3 CNXyl CNXyl
19F NMR (δ/ppm)
δ -75.16 ppm (s, SO2CF3)
1H NMR (δ/ppm)
δ 2.29 ppm (s, 12H, CH3 of XylNC) δ 2.36 ppm (s, 6H, CH3CN) δ 5.22 ppm (s, 2H, CHPhCHPh)
IR (KBr): 2121 cm-1 XylNC = 2,6-(CH3)2C6H3NC 24 h Ru N N Tf Tf C6H5 C6H5 NCCH3 NCCH3 NCCH3 NCCH3 N N Ru N N Tf Tf C6H5 C6H5 NCCH3 NCCH3 N 1.95, 2.63 (bs, s, 3H, CH3CN)
- 77.17, -75.43 (CF3, each s)
N single diastereomer (1.0 equiv.) ClCH2CH2Cl, 60°C 86% N N N N = Ru N N Tf Tf C6H5 C6H5 N NCCH3 N NCCH3
∆ Λ
1H NMR (δ/ppm)
19F NMR (δ/ppm)
- r
5.11, 5.81 (s, 2H, NHPhNHPh) 4 days 05
SLIDE 6
Pd(OAc)2 Pd R N N R NCCH3 NCCH3 + CH3CN, reflux pale yellow
Preparation of N,N'-bis(sulfonylamido)Pd(II) Complexes
+ 2 AcOH 24 h 81% 32% 80% 66% NH NH C6H5 C6H5 R R Pd N N NCCH3 NCCH3 Pd N N NCCH3 NCCH3 Pd N N NCCH3 NCCH3 Pd N N NCCH3 NCCH3 Nf = n-C4F9SO2- Pf = C6F5SO2- C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 C6H5 Tf Tf Ts Ts Nf Nf Pf Pf 1.0 equiv. 06
SLIDE 7
Pd N N solvent, rt
Reaction of Pd Complex with Various Ligands
Characterized by 1H and 19F NMR. aIn the presence of MS 4A. L L2 equiv. yield, % t-BuNC 1.5 1.5 99 98 96 colorless 2.1 N N 99 1.0 solvent THF THF THFa CH2Cl2 pale yellow NCCH3 NCCH3 Pd N N L L
- 2CH3CN
C6H5 C6H5 C6H5 C6H5 Tf Tf Tf Tf
07
SLIDE 8
Rearrangement of Allyl Ester via Kinetic Resolution
Pd cat C6H5 O O Pd cat conv, %a Pd[(S,S)-Ts2dpen](CH3CN)2 59 62 Conditions; [ester] = 2.0 x 10-1 M.
aDetermined by GC (CHIRASIL DEX-CB) and n-dodecane as an internal standard.
Pd[(S,S)-Nf2dpen](CH3CN)2 Pd[(S,S)-Pf2dpen](CH3CN)2 3 ee, %a kS/kR
b
Pd[(S,S)-Tf2dpen](CH3CN)2 48 43.7 0.3 48.0 60.5 1.0 2.5 2.1 4.3 kS/kR = ln (1-conv)(1- ee) ln (1-conv)(1+ee)
bCalculated based on the equation shown below.
C6H5 O O C6H5 O O S R C6H5 O O + R + 08 60 °C, 8 h ClCH2CH2Cl S/C = 20
SLIDE 9
SUMMARY
N Pd N NCCH3 NCCH3 SO2 R SO2 R N Ru N SO2 R SO2 R NCCH3 NCCH3 NCCH3 NCCH3 Pd cat C6H5 O O C6H5 O O C6H5 O O S R C6H5 O O + R + N Ru N SO2 R SO2 R NCCH3 NCCH3 NCCH3 NCCH3 L
- CH3CN