SLIDE 1
1 Cp*Ru(PN) CATALYSTS WITH A BIFUNCTION BASED ON METAL/NH - - PowerPoint PPT Presentation
1 Cp*Ru(PN) CATALYSTS WITH A BIFUNCTION BASED ON METAL/NH - - PowerPoint PPT Presentation
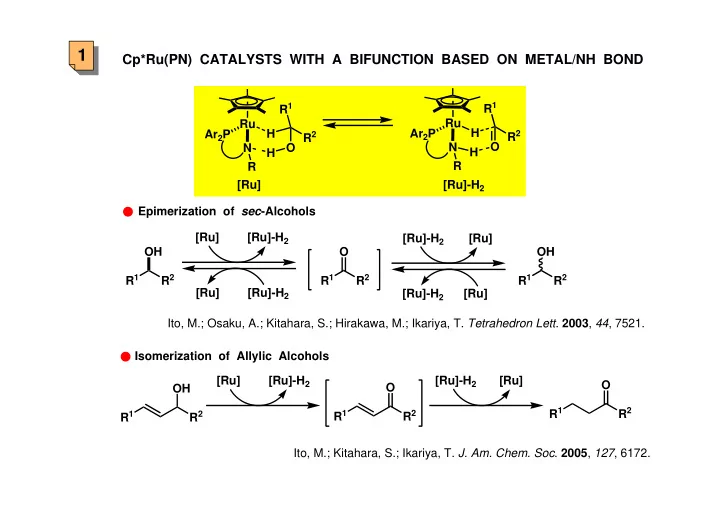
1 Cp*Ru(PN) CATALYSTS WITH A BIFUNCTION BASED ON METAL/NH BOND R 1 R 1 Ru Ru H H Ar 2 P Ar 2 P R 2 R 2 O O N N H H R R [Ru] [Ru]-H 2 Epimerization of sec -Alcohols [Ru] [Ru]-H 2 [Ru]-H 2 [Ru] OH O OH R 1 R 2 R 1 R 2 R 1 R
SLIDE 2
SLIDE 3
POSSIBLE MECHANISM FOR LACTONIZATION WITH Cp*Ru CATALYST
3
Ru NH P O OH O OH O O OH O O OH OH OH Ru Cl P N H H base
- HCl
OH OH + O 2 O O OH 2 + Ru cat KOt-C4H9 acetone R R R R Ar Ar Ar Ar Ru H P N H H Ar Ar Ru H P N H H Ar Ar Ru NH P Ar Ar
SLIDE 4
REGIOSELECTIVE LACTONIZATION OF UNSYMMETRICAL 1,4-DIOLS
4
OH OH O O Ru cat KOt-C4H9 acetone R R O + R O
1 2 4 4 1 2 4 1 2
OH OH O Ru cat KOt-C4H9 acetone + OH O OH OH Ru cat KOt-C4H9 acetone +
n
O OH O
n n
2-Substituted-1,4-diols 4-Substituted-1,4-diols 4-Substituted-triols R O R
4 1
(n = 1, 2, 3)
4 1 4 1 4 1
R
4 1
O O
4 1
HO HO
SLIDE 5
Kamlage, S.; Sefkow, M.; Zimmermann, N.; Peter, G. M. Synlett 2002, 77.
REPORTED REGIOSELECTIVE LACTONIZATION OF 2-SUBSTITUTED-1,4-BUTANEDIOLS
5
Ar OH OH toluene 20 °C , 10 h O Ph + Ar O O O Ar O + + O Ph 3,4-dimethoxyphenyl A B Ar yield (%) A:B 79 95:5 3,4-methylenedioxyphenyl 75 96:4 Shao, L.; Miyata, S.; Muramatsu, H.; Kawano, H.; Ishii, Y.; Saburi, M.; Uchida, Y.
- J. Chem. Soc., Perkin Trans. 1, 1990, 1441.
RuH2(PPh3)4 2 2 S/C = 25 Ar OH OH + O N O CH3 CH2Cl2 Ar O O + Ar O O (n-C3H7)4RuO4 67 - 82% yield Ar = phenyl or 3,4-methylenedioxyphenyl 67:33
SLIDE 6
REPORTED REGIOSELECTIVE LACTONIZATION OF POLYOLS
6
HO OH OH acetone 180 ゜ C, 3 h O HO O Murahashi, S.; Naota, T.; Ito, K.; Maeda, Y.; Taki, H. J. Org. Chem. 1987, 52, 4319. 71% RuCl2(PPh3)3 S/C = 20
1 2 6 1 2 6
+ 2 + 2 O OH HO OH OH OH OH OH O O HO HO HO OH 90% DMF 80 ゜ C RhH(PPh3)4 O Ph + 2 O Ph + 2 S/C = 2.5 Isaac, I.; Aizel, G.; Stasik, I.; Wadouachi, A.; Beaupere, D. Synlett 1998, 475.
SLIDE 7
SYNTHESIS OF 2-ARYLMETHYL-1,4-DIOLS
7
Itooka, R.; Iguchi,Y.; Miyaura, N. J. Org. Chem. 2003, 68, 6000. [RhCl(cod)]2 N(C2H5)3, 1.0 equiv 1,4-dioxane, H2O (6/1) rt OH OH LiAlH4, 3.0 equiv THF, reflux Ar O O CH3O yield, % (2 steps) 92 73 91 + Rn B(OH)2 Rn Rn PhCH2O 70 OCH3 OCH3 O O OCH3 OCH3 O O 1.1 equiv S/C = 33
SLIDE 8
LACTONIZATION OF 2-BENZYL-1,4-BUTANEDIOL CATALYST TUNING: COUNTER ANION AND Cp STRUCTURES
8
Ru NCCH3 Ph2 P N H2 77:23 77:23 OTf Ru NCCH3 Ph2 P N H2 79:21 Cl OH OH Ph Ru cat KOt-C4H9 acetone 30 °C, 1 h A B Ph O O Ph O O + S/C = 100, Ru cat:KOt-C4H9 = 1:1 Ru cat: >99% yield A:B* Ru NCCH3 Ph2 P N H2 PF6 *determined by 1H NMR analysis 56:44 [diol] = 0.5 M Ru Cl Ph2 P N H2
SLIDE 9
LIGAND EFFECT: N- ,P- AND N-α-SUBSTITUENTS
9
A:B Ru cat 78:22 82:18 83:17 Ar2P N 81:19 76:24 78:22 81:19 (4-tol)2 (2,6-xy)2 A:B Ru NCCH3 P N OTf NH2 Ph2P NHCH3 Ph2P NHCH2Ph Ph2P NH2 Ph2P Ph NH2 Ph2P CH2Ph NH2 P CH2Ph NH2 P CH2Ph 4-tol 2,6-xy Ph Ar: Ar Ar Ar2P N
SLIDE 10
LIGAND EFFECT: BACKBONE
10
OH OH Ph Ru cat KOt-C4H9 acetone 30 °C, 1 h A B Ph O O Ph O O + S/C = 100, Ru cat:KOt-C4H9 = 1:1 [diol] = 0.5 M A:B P N 92:8 P NH2 Ph2 82:18 P NH2 Ph2 92:8 92:8 P NCH3 Ph2 P NCH2Ph Ph2 H H 47 20 yield, % >99 >99 Ru cat Ru NCCH3 P N OTf 85 (0 °C) 93:7
SLIDE 11
EFFECT OF ARYLMETHYL SUBSTITUENTS IN 1,4-DIOL
11
A:B 92:8 92:8 92:8 NCCH3 Ph2 P N H2 OTf Ru cat S/C = 100, Ru cat:KOt-C4H9 = 1:1 [diol] = 0.5 M 92:8 Ru OH OH OH OH OH OH OH OH A:B yield, % >99 >99 >99 >99 yield, % O O PhCH2O CH3O
SLIDE 12
1) LDA, THF/HMPA 2) O O Br O O O O O Hinokinin 1) LHMDS, THF 2) CHO CH3O CH3O CH3O O O CH3O OCH3 OCH3 O O Isodeoxypodophyllotoxin Bode, J. W.; Doyle, M. P.; Protopopova, M. N.; Zhou, Q. -L. J. Org. Chem. 1996, 61, 9146. O O O O O 3) TFA
12
REPORTED SYNTHESIS OF BIOACTIVE LIGNANS
SLIDE 13
SYNTHESIS OF JUSTICIDIN E
13
Ru cat KOt-C4H9 acetone 30 °C, 15 h S/C = 50, Ru cat:KOt-C4H9 = 1:1 OH OH O O O O O O O O O O O O O O O O 94% 0% Justicidin E Taiwanin C + NCCH3 Ph2 P N H2 OTf Ru cat Ru
SLIDE 14
LACTONIZATION OF UNSYMMETRICAL 4-SUBSTITUTED-1,4-DIOLS
14
diol:Ru cat:KOt-C4H9 = 100:1:1 [diol] = 0.5 M + O 2 Ru cat KOt-C4H9 30 °C, 1 h + OH 2 acetone The 79th Annual Meeting of Chemical Society of Japan. 2001, 1H326 R1 R2 yield, % CH3 CH3 H D (96%) >99 >99 (96% atom D) diol:Ru cat:KOt-C4H9 = 100:1:1 [diol] = 0.5 M OH OH + O 2 Ru cat KOt-C4H9 30 °C, 1 h O + OH 2 acetone >99% primary vs tertiary primary vs secondary CH3 CH3 O CH3 CH3
1 4 1 4
OH OH R1 R2
1 4
O O R1 R2
1 4
Cl Ph2 P N H2 Ru Ru cat
SLIDE 15
LACTONIZATION OF TRIOLS
15
Cl Ph2 P N H2 Ru Ru cat OH OH O O OH OH O O HO O O HO OH OH OH O O OH O O HO Ru cat KOt-C4H9 acetone 30 °C, 31 h diol:Ru cat:KOt-C4H9 = 33:1:1 [diol] = 0.1 M Ru cat KOt-C4H9 acetone 30 °C, 48 h diol:Ru cat:KOt-C4H9 = 33:1:1 [diol] = 0.1 M >99% >99%
1 4 6 1 4 6 1 4 6 1 4 6 1 4 7 1 4 7 1 4 7
0% 0% 0% Lactonization of 1,4,6-triol Lactonization of 1,4,7-triol
SLIDE 16
LACTONIZATION OF 1,4,5-TRIOLS: SYNTHESIS OF L-FACTOR AND MURICATACIN
16
R = C5H11 [triol] = 0.5 M R = C12H25 [triol] = 0.05 M KOt-C4H9 Ru cat acetone 30 ゜ C, 1 h O O R OH triol:Ru cat = 100:1 >99% Ru Cl Ph2 P N H2 Ru cat O O HO O O HO L-Factor muricatacin Autoregulatory factor of the antibiotic-producing Gram-positive actinomycete Streptomyces griseus Obtained from Annona muricate Potent cytotoxic agent on tumor cell lines OH OH R OH
1 4 5 1 4 5
SLIDE 17