SLIDE 1
- A. Chromium Based Reagents
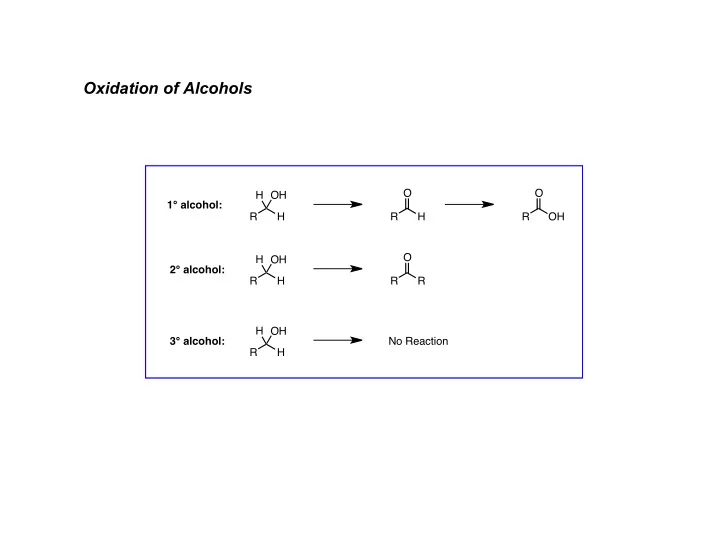
General Mechanism:
- 1° alcohols: under anhydrous conditions (Collins, PCC, PDC) will stop at aldehyde
- in presence of aqueous acid (Jones), see further (rapid) oxidation to carboxylic acid
- oxidation of 2° alcohols give ketones
- these processes generate chromium waste (toxic)