SLIDE 1
RINGOPENING OF EPOXIDES WITH ALCOHOLS R H H C R O H + - - PowerPoint PPT Presentation
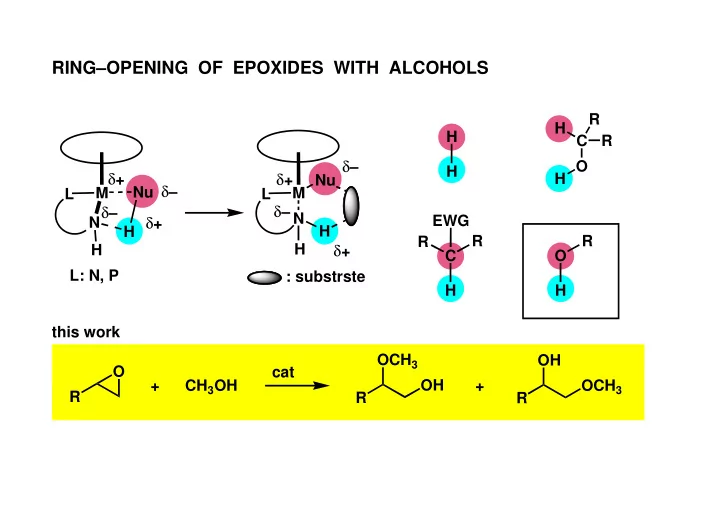
RINGOPENING OF EPOXIDES WITH ALCOHOLS R H H C R O H + - - PowerPoint PPT Presentation
RINGOPENING OF EPOXIDES WITH ALCOHOLS R H H C R O H + + H Nu Nu M M L L + N EWG N H H R R R + H H C O L: N, P : substrste H H this work OCH 3 OH O cat + CH 3 OH OH +
SLIDE 2
SLIDE 3
C6H5 + ROH epoxide:ROH:cat = 50:500:1 50 °C, 24 h
REACTION OF STYRENE OXIDE WITH ALCOHOLS
Ru cat (CH3)3COH C6H5 C6H5 OR OH + OH OR A B CH3OH ROH Ru cat: Ru[(S,S)-Tsdpen](p-cymene) CH3CH2OH yield, % A byproducts B byproducts: C6H5 OH C6H5 OH 26 1 <1 <1 22 1 34 25 O ROH
SLIDE 4
LIMITATION
C6H5 C6H5 C6H5 O O O O O reactive inert inert inert
SLIDE 5
REACTION OF rac-STYRENE OXIDE WITH METHANOL
O epoxide:CH3OH:cat = 50:500:1 CH3OH OCH3 OH OH OCH3 + 95:5 * * 20% yield 68% ee (R) racemate 34% conv 50 °C, 6 h CH3OH (S,S)-Ru cat: Ru[(S,S)-Tsdpen](p-cymene) (S,S)-Ru cat + N H Ru N Ts C6H5 C6H5 A B
SLIDE 6
REACTION OF (S)-STYRENE OXIDE WITH METHANOL
O 50 °C, 24 h epoxide:CH3OH:cat = 50:500:1 + OCH3 OH A Ru cat Ru[(S,S)-Tsdpen](p-cymene) Ru[(R,R)-Tsdpen](p-cymene) Ru(Ts-en)(p-cymene)
S
>99% ee CH3OH yield, % ee, % (config) yield, % product (A) recovered epoxide
R
Ru cat CH3OH OH OCH3 + B * 9 6 (R) 58 67 95 (S) 97 (S) 68 31 27 40 (R) 76 (R) 63 (R) ee, % (config)
SLIDE 7
O solvent epoxide:solvent:chiral cat = 50:500:1 50 °C, 24 h
S
O chiral Ru cat C6H5 N H Ru N Ts Ts = p-CH3C6H4SO2 solvent ee, % (config) yield, % CH3OH C6H6 (CH3)3COH chiral Ru cat: C6H5 * * chiral Ru cat
RACEMIZATION OF (S)-STYRENE OXIDE
S,S R,R CH3OH C6H6 (CH3)3COH S,S R,R S,S R,R 9 58 95 97 >99 >99 5 (R) 97 (S) 14 (R) 99 (S) 57 (R) 20 (R) recoverd epoxide
SLIDE 8
O cis epoxide:CH3OH:Ru= 50:500:1 O cis trans O (±)-trans O CH3OH 50 °C, 24 h CH3OH 50 °C, 24 h 14% yield 19% ee
cis–trans ISOMERIZATION OF STILBENE OXIDE
no reaction 14% conv (S,S)-Ru cat (S,S)-Ru cat (S,S)-Ru cat: Ru[(S,S)-Tsdpen](p-cymene)
SLIDE 9