SLIDE 1
Constants and units
- Charge on an electron exactly equals that of a proton (1.602x10-19 Coulombs)
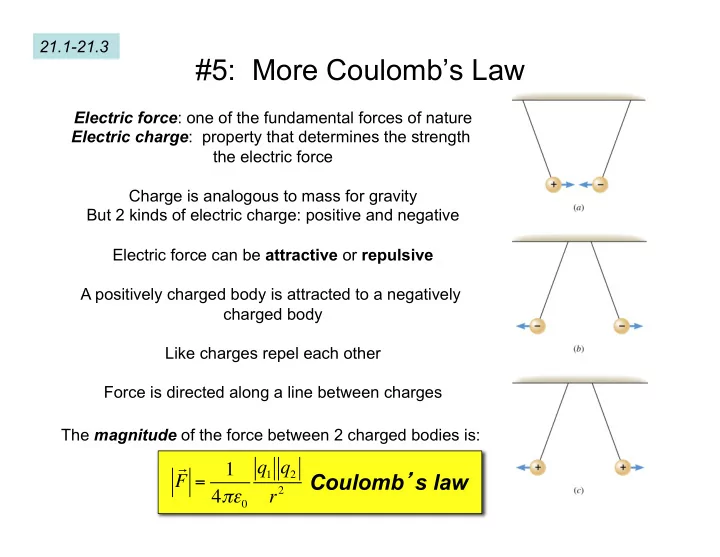
#5: More Coulombs Law Electric force : one of the fundamental forces - - PowerPoint PPT Presentation
21.1-21.3 #5: More Coulombs Law Electric force : one of the fundamental forces of nature Electric charge : property that determines the strength the electric force Charge is analogous to mass for gravity But 2 kinds of electric charge:
12
q1 ¡ q2 ¡
12
12
F
12
F
21
q1 ¡ q2 ¡
r
12
ˆ r
12
F
12
F
21
12
12 =
12 2
12
12
1 =
12 +
13 +
14 +
1C 2C 1C 1m 2m
2C 1C 1m 2m
q1 q2
2
2
2