SLIDE 1
Working principle of WP4 Block chamber Works on relative humidity - - PowerPoint PPT Presentation
Working principle of WP4 Block chamber Works on relative humidity - - PowerPoint PPT Presentation
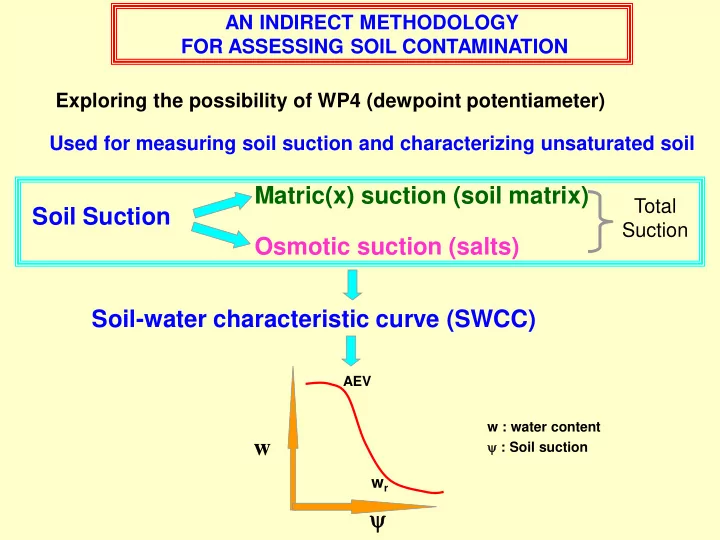
AN INDIRECT METHODOLOGY FOR ASSESSING SOIL CONTAMINATION Exploring the possibility of WP4 (dewpoint potentiameter) Used for measuring soil suction and characterizing unsaturated soil Matric(x) suction (soil matrix) Total Soil Suction Suction
SLIDE 2
SLIDE 3
A Case study
Soil used: Marine soil designated as contaminated soil (CS) Source: Collected from the coastal area of Mumbai, India
Soil property Value Specific gravity 2.64 Particle size characteristics Coarse sand (4.75-2.0 mm) 4 Medium sand (2.0-0.420 mm) 9 Fine sand (0.420-0.074mm) 11 Silt size (0.074-0.002 mm) 44 Clay size (< 0.002 mm) 32 Consistency limits Liquid limit (%) 61 Plastic limit (%) 37 Plasticity index (%) 24 Soil Classification (USCS) MH Oxide % by weight SiO2 33 Al2O3 11 Fe2O3 12 TiO2 2 CaO 6 Chlorides (ppm) 9840 Sulphites (ppm) 40 CEC (meq/100g) 4.04
Physical properties Chemical properties As such the soil is contaminated
SLIDE 4
Soil subjected to washing to nullify contamination
- No. of washings
LS Chloride (ppm) Sulphite (ppm) 1 2 6750 15 2 4 1850 10 3 6 800 10 4 8 250 5 5 10 90 < 5
1 2 3 4 5 6 7 10 20 30 40 50 60 70 80 90 100 (ms/cm)
- No. of washings
Washing nullifies contamination 10 10
1
10
2
10
3
10
4
10
5
10
6
10
7
0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 (kPa)
w
Contaminated soil Washed soil Difference due to contamination
SLIDE 5
For geotechnical engineers, it’s very important subject Metal corrosion in undisturbed soils is generally very low regardless of the soil composition (e.g. metal piles, reinforcement of foundation etc.) Corrosion of metal (steel) in disturbed soils (e.g., buried pipelines that are backfilled) is strongly affected by soil conditions & properties. Soil changes its chemical and physical nature continuously over time and seasonally.
Corrosion Potential of Soils
Pipeline damage from pitting/corrosion
SLIDE 6
- Chloride content
- Moisture content
- Oxygen content/Redox potential
- Soil permeability/texture
- pH/Acidity
- Temperature
- Soil resistivity
- Drainage characteristics
- Sulfate/Sulfite ion concentrations
- Microbiological activity
- Stray currents (from cathodic protection, DC traction
system viz., train, metro)
- Spillage of corrosive substance/pollution
Soil Characteristics & Environmental Variables
SLIDE 7
Clay in the soil mass reduces movement of air (oxygen) and water, i.e. low aeration, when wet, and hence increase in local (pitting) corrosion. High plasticity of clay (swelling/shrinking soils) can pull off susceptible coatings on the structures. Clay is susceptible to cracking (during wetting-drying cycles) which helps transport of air and moisture to the structures buried in it. Sand promotes aeration and moisture distribution & hence, soluble salts and gases (air/oxygen) are easily transported to structures, causing greater general corrosion but less pitting.
Soil Classification/Texture
SLIDE 8
Bored Cast in-situ piles
Reinforcement in concrete pile exposed due to leaching of concrete Chloride and Sulphate content of water found well within prescribed limit & hence water not corrosive. Ryzner index (RI) of water was found out to be 7.7 & hence water is corrosive and unsaturated
SLIDE 9
pH scale for Soils
Langelier Saturation Index (LI)
Determines if calcium carbonate will precipitate or not LI = pH – pHs pH = actual pH value measured in the water pHs = pH of the water in equilibrium with solid CaCO3 If LI > 0 calcium carbonate will precipitate If LI < 0 calcium carbonate won’t precipitate The CaCO3 layer deposited on surfaces acts as a protective coating.
Ryznar Index
Determines the degree of scale formation RI = 2 pHs – pH RI < 5.5 heavy scale will form 5.5 < RI < 6.2 scale will form 6.8 < RI < 8.5 water is corrosive RI > 8.5 water is very corrosive
SLIDE 10
ASSESSMENT OF CORROSION POTENTIAL OF SOILS
Durability of underground structures is seriously affected by corrosion of the concrete (IS: 456-2000) Specifications for type of cement, minimum cement content, maximum water-cement ratio, etc., to be adopted stringently, based on the exposure
- f the concrete to different concentrations of sulphates in the soil or
ground water. However, for assessment of corrosion potential of underground structures, chemical properties of the soil need to be considered in details. Corrosion is an electrochemical process Certain conditions must exist for the corrosion to occur (corrosion cell)
Effects of soil characteristics on corrosion By Victor Chaker, J. David Palmer ASTM Committee G-1 on Corrosion of Metals
SLIDE 11
Soil (Electrolyte) Metallic connection Anode Cathode Electric current
Electrochemical reaction
Corrosion
The “Corrosion cell”
Soil Electrolyte
Therefore properties of soils play a crucial role in accelerating corrosion.
Properties of soils:
Electrical resistivity pH moisture content Porosity sulphate and chlorides content redox potential presence of micro-organism temperature are important for evaluating the corrosion potential of soils (DIN 50929-3). For corrosion, the elements that are soluble in water are important: – Base forming: Na, K, Ca, Mg (raise pH). – Acid forming: Carbonate, Bicarbonate, Chloride ion, Nitrate, and Sulfate (lower pH).
SLIDE 12
Rating based on the soil fraction Rating based on the electrical resistivity Rating based on the pH Rating Based on the ground water status Rating based on the sulphite content Rating based on the chloride content
Based on different soil characteristics, a certain rating (R1 to R6) for the soils has been assigned and the sum of these ratings is a measure of the overall soil corrosivity.
SLIDE 13
Rating based on the soil fraction
Soil fraction % by weight R1 Clay & silt <10 +4 10 to 30 +2 30 to 50 50 to 80
- 2
>80
- 4
Organic matter, e.g.: muddy or swampy soils: peat, mud, marsh >5
- 12
Severely polluted: due to fuel ash, slag coal, coke, refuse, rubbish or waste water
- 12
Rating based on the electrical resistivity Resistivity (.m) R2 >500 +4 200 to 500 +2 50 to 200 20 to 50
- 2
10 to 20
- 4
<10
- 6
Rating based on the pH PH R3 >9 +2 5.5 to 9 4.0 to 5.5
- 1
<4
- 3
Higher conductivity: high corrosion rate (efficient electrolyte)
SLIDE 14
Rating Based on the ground water status Ground water status R4 No groundwater Groundwater
- 1
Groundwater at times -2 Rating based on the sulphite content Sulphite content (g/l) R5 <0.15 0.15 to 1
- 2
1 to 2
- 4
>2
- 6
Rating based on the chloride content Chloride content (ppm) R6 <100 100-2000
- 2
2000-10000
- 4
>10000
- 6
Total assessment of the corrosion potential Summation of R1- R6 R Corrosion potential 0 Virtually not corrosive
- 1 to -4
Slightly corrosive
- 5 to -10
Corrosive < -10 Highly corrosive
Chloride ions: Cause pitting of steel and decrease soil resistivity.
SLIDE 15
Dissolved Oxygen concentration in the soil moisture determines its RP(potential
- diff. between the electrodes), higher the oxygen content, higher would be the RP