The Reaction Between Captured CO2 and NaOH Produces Na2CO3 For Use - PowerPoint PPT Presentation
The Reaction Between Captured CO2 and NaOH Produces Na2CO3 For Use In Solar Cells T eam Members: Sabiha Haque, Nora Heaphy, Owen Heaphy, Daniel Huang, Kenechi Nkwo, and Laura Wainman Mentor: Harishankar Jayakumar Problem Carbon dioxide
The Reaction Between Captured CO2 and NaOH Produces Na2CO3 For Use In Solar Cells T eam Members: Sabiha Haque, Nora Heaphy, Owen Heaphy, Daniel Huang, Kenechi Nkwo, and Laura Wainman Mentor: Harishankar Jayakumar
Problem ● Carbon dioxide emissions contribute to climate change Alternative energy sources like ● solar power are critical to reducing CO2 emissions Solar power is underused due to ● its cost and intermittency ● Carbon capture & sequestration methods are inefficient and unsustainable
Background Our team researched international INDCs (Intended ● Nationally Determined Contributions) under the Paris Agreement. We then looked into the carbon capture process within ● large factories. ● The members our team with chemistry expertise suggested using a fairly common reaction, but in an innovative way, to change harmful CO2 emissions into Na2CO3 .
Hypothesis If CO2 emissions are captured and reacted with NaOH compound, their reaction will produce Na2CO3, a compound that can be used to produce solar cells. H0: μ1 - μ2 = 0 Ha: μ1 - μ2 > 0, where μ1 = final benefit and μ2 = initial cost
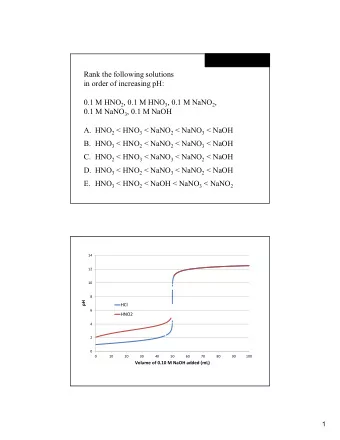
Methods: ● Tested the effects of atmospheric absorption on aqueous NaOH ● Performed a titration ● Exposed the NaOH solution to the atmosphere over the course of 24 hours ● Used KHP as a standard titration agent ● Used stoichiometry to find theoretical yield of products
Video of Lab
Lab Analysis/Results: ● Average molarity of NaOH for day #1 is 1.06 M and is 0.926 M for day #2 ● Amount of Na2CO3 produced for 1.06 M of NaOH is 28.1g, and the amount produced for 0.926 M is 24.5g ● Over a period of a day, the amount of Na2CO3 produced and NaOH consumed decreased by 12.6%
Potential Lab and Reaction Errors: ● Crystallization: separation of aqueous Na2CO3 from liquid H2O can lead to impurities and can be expensive Impurities: may have impurities and separate reactions may have happened simultaneously, ● producing other products ● Simultaneous Reactions: different reactions going on simultaneously may affect how accurate our data is, and how the reaction can be implemented in factories Absorption of NaOH: in implementation of the reaction, the molarity of NaOH will decrease ● due to atmospheric absorption, lowering its efficacy These will need to be further tested and controlled for in regards to future testing. Additionally, we will need to look at how this reaction will take place in a model and what other reactions may occur simultaneously, as well as whether these simultaneous reactions produce toxic products.
Solution ● Convert the chemical reaction of NaOH and CO2 into technology that can be implemented within smokestacks to transfer the captured CO2 into Na2CO3 Utilize the captured Na2CO3 as a resource for increasing solar cell ● manufacturing (ultimately lessening CO2 emissions by recycling them)
Printed 3D Model
Process Specifications For our desulfurisation process, noting that the wet desulfurization we used will produce the suspension of the pool and the reaction of the secondary entrainment, resulting in excessive dust emissions, in order to attain the standard of desulfurisation and dust controlling, the processing part should contain 7 major factors : 1. contained efficient demisting agent; 2. the reactants should be changed 3.contained filtration device ; 4. contained aeration device ; 5. size of pool should be large in the standard of 120m^3 per pool for every 10,000 m^3/h of flue gas ; 6. composite flocculant is used ; 7. denitrification device is installed. (Data calculations and device suggestion credits to Professor Wu from Hejing Environmental Protection in Foshan City, China)
Next Steps ● Conduct a cost-benefit analysis of potential economic viability of our solution ● Conduct market research and survey potential customers Further pursue project within the Sustainable Development Goals Challenge ● Questions for future research: ● ○ How can we perform this reaction effectively on an industrial scale? What is the final product likely to cost, both for materials and equipment? ○ ○ What apparatus will create the best installment of our solution in smokestacks? How can we verify the sustainability of the solar cells fueled by Na2CO3? ○ ○ Since our reaction is also a large scale of exothermic reaction, can we also reuse the heat that has been wasted during the reaction? Create a mathematically-equivalent model of the process ● ● Develop a prototype for engineering and design
Citations Ashford, Molika. (2008, Oct 1). A better CO2 scrubber. Retrieved from http://www.popsci.com/molika-ashford/article/2008-10/better-co2-scrubber Ge, Zhiwei, et al. (2013, Sep 2). Carbonate-salt-based composite materials for medium and high temperature thermal energy storage. Retrieved from http://www.sciencedirect.com/science/article/pii/S1674200113001958 Science Encyclopedia. (2016). Electrolysis - production of sodium hydroxide, chlorine, and hydrogen. Retrieved from http://science.jrank.org/pages/2351/Electrolysis-Production-sodium-hydroxide-chlorine-hydrogen.html Stolaroff, Joshuah, et al. (2008). Carbon dioxide capture from atmospheric air using sodium hydroxide spray. Retrieved from http://scholar.harvard.edu/files/davidkeith/files/97.stolaroff.aircapturecontactor.e.pdf Wee, Jung-Ho, et al. (2011, Jan). A comparison of solar voltaics and molten carbonate fuel cells as commercial power plants. Retrieved from http://www.sciencedirect.com/science/article/pii/S1364032110002996 Zeman, Frank, et al. (2004). Capturing carbon dioxide directly from the atmosphere. Retrieved from https://pdfs.semanticscholar.org/1dae/d9728aed8e6c1e344b4aa387abddf271ccc9.pdf Hejing Environmental Protection Co,.td. (2014) 陶瓷 废 气深化治理 ( Deepening Treatment Technology of emission of ceramic production )
Thank You We would like to thank the following for his or her help in our project… Dr. Harishankar Jayakumar (mentor), from City College of New York ● ● Mr. George Householder, from Glastonbury High School ● Dr. Nazia Mintz Habib, from Cambridge University Professor Wu Rongbiao, from Hejing Environmental Protection( 合璟 环 保) ● Mr. Andy Bauer, from Glastonbury High School ●
Recommend
More recommend
Explore More Topics
Stay informed with curated content and fresh updates.