1
Rank the following solutions in order of increasing pH: 0.1 M HNO2, 0.1 M HNO3, 0.1 M NaNO2, 0.1 M NaNO3, 0.1 M NaOH
- A. HNO2 < HNO3 < NaNO2 < NaNO3 < NaOH
- B. HNO3 < HNO2 < NaNO2 < NaNO3 < NaOH
- C. HNO2 < HNO3 < NaNO3 < NaNO2 < NaOH
- D. HNO3 < HNO2 < NaNO3 < NaNO2 < NaOH
- E. HNO3 < HNO2 < NaOH < NaNO3 < NaNO2
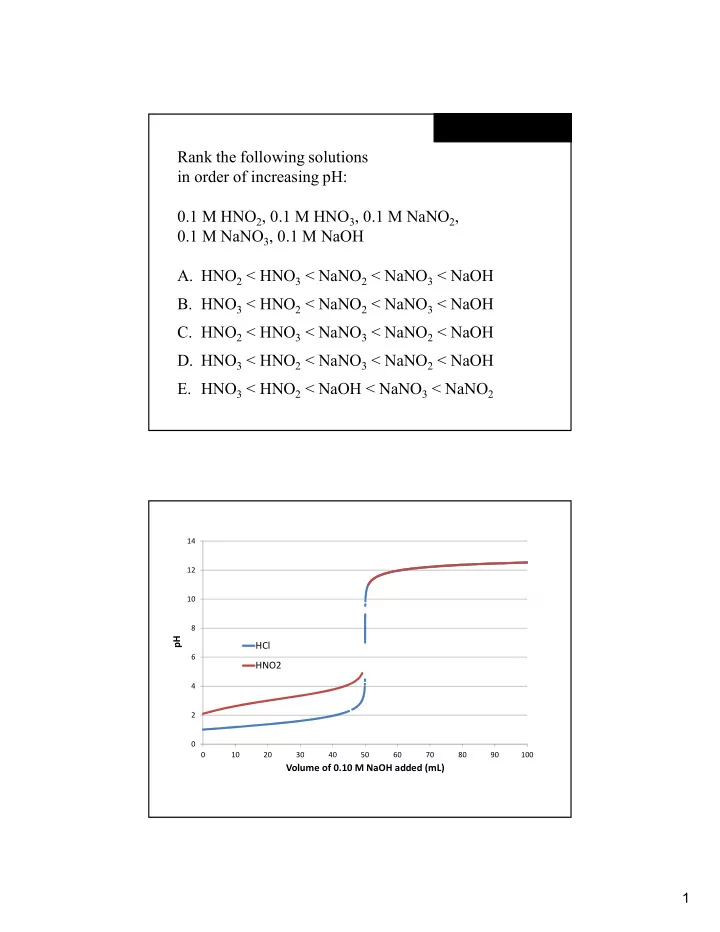
2 4 6 8 10 12 14 10 20 30 40 50 60 70 80 90 100
pH Volume of 0.10 M NaOH added (mL) HCl HNO2