Oxidation of Ni-10Cr-9Al alloy at 1000 0 C Pressure Dependence of SO - - PowerPoint PPT Presentation
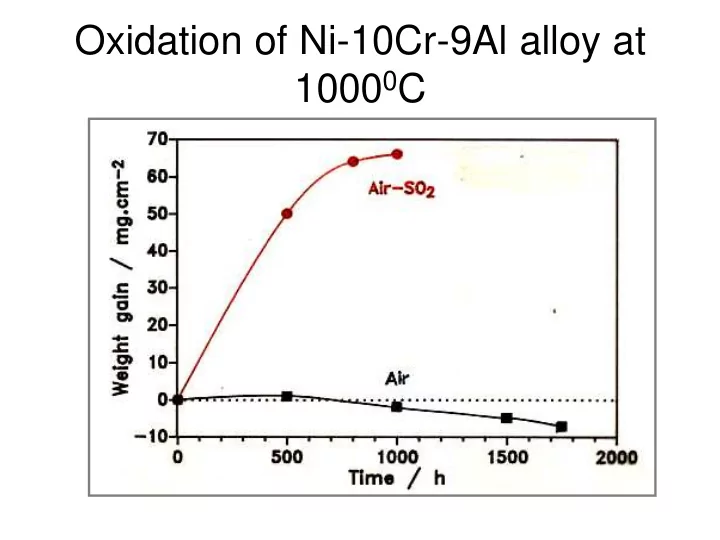
Oxidation of Ni-10Cr-9Al alloy at 1000 0 C Pressure Dependence of SO - - PowerPoint PPT Presentation
Oxidation of Ni-10Cr-9Al alloy at 1000 0 C Pressure Dependence of SO 2 Reaction with SO 2 is tempertaure and Pressure Dependent At 1 atms. SO 2 pressure at 500-600 o C 7Ni + SO 2 = 4NiO + Ni 3 S 2 ------- [1] At this pressure as per Phase
Pressure Dependence of SO2
- Reaction with SO2 is tempertaure and Pressure
Dependent
- At 1 atms. SO2 pressure at 500-600oC
7Ni + SO2 = 4NiO + Ni3S2 ------- [1]
At this pressure as per Phase stability Diagram, NiSO4 is also stable, because of which there is additional reaction
9Ni + NiSO4 = 8NiO + Ni3S2 ------- [2]
Because of which the rate of reaction is very fast. As the pressure is reduced from 1 atm to 0.1 atms. NiSO4 is no longer stable and reaction 2 does not occur and rate falls.
Phase Stability Diagram Of Ni
Temperature Dependence of SO2
- NiSO4 is stable upto 600oC, hence no possibility of
reaction [2].
- At 635oC Ni-S liquid starts becoming stable. The reaction
from 700-900oC are linear and rates are proportional to SO2 pressure.
- The rate decreases further with increase in temp. above
800oC because of instability of Ni3S2 above 800oC.
- The sulphide formation becomes thermodynamically
unstable at high temperatures and at low SO2 pressure
- Under these conditions, the corrosion product is is only
NiO and no sulphide thus corrosion rate falls drastically.
- That is why rate of corrosion of Ni in SO2 at 1000oC is
almost same as rate of oxidation of Ni.
Sulphidation of Ni Vs Co
Sulphidation of Ni in SO2 + O2 NiO (s) + SO3 (g) = NiSO4 (s) SO2 (g) + O2 (g) = SO3 (g)
Oxidation of Binary Alloys of Ni-Cr
Sulphidation of Fe-Cr, Ni-Cr and Co-Cr Alloys
Effect of Pre-Oxidation on Sulphidation
Mechanism of SO2/SO3 Reaction
- Reaction starts with the sulphide formation of most stable element
and further growth occurs mainly by lattice diffusion of ions thru sulphide scales.
- Formation of mixed oxide scale occurs even when the partial
pressure of oxygen or sulphur is dominant at one particular point. – this happens because of continuous increase in conc. Of other gas below the main scale interface.
- How the sulphur partial pressure in craeses below oxide scale
(a) Dissolution of SO2 – not sound as s disolution is very low. (b) Gas dissolution thru’ pores and cracks and hence establishes con. Gradient. (c )Direct reaction of SO2 which can produce S C + SO2 = S + CO2