Professional Publications, Inc.
FERC
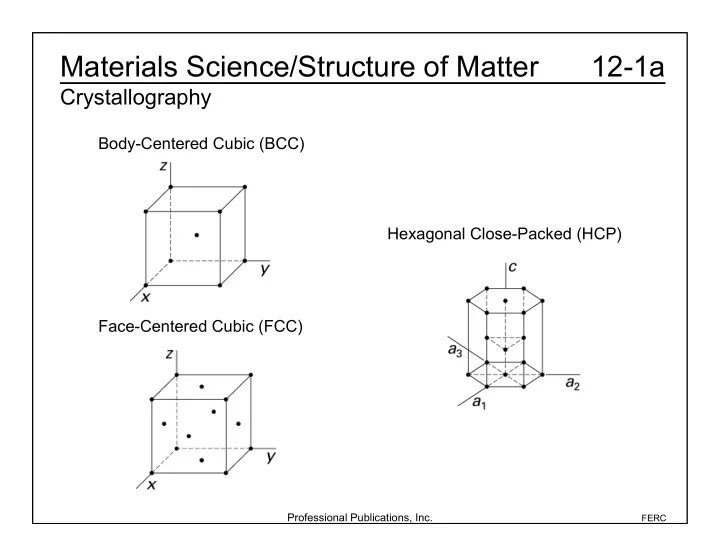
Materials Science/Structure of Matter 12-1a Crystallography - - PowerPoint PPT Presentation
Materials Science/Structure of Matter 12-1a Crystallography Body-Centered Cubic (BCC) Hexagonal Close-Packed (HCP) Face-Centered Cubic (FCC) Professional Publications, Inc. FERC Materials Science/Structure of Matter 12-1b Crystallography
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
1 4 1 4 3 4
_
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
0 M n+ + ne
2 O2 + 2e +H2O 2OH 1 2 O2 + 2e + 2H3O + 3H2O
+ 2H3O + 2H2O+H2
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC
Professional Publications, Inc.
FERC