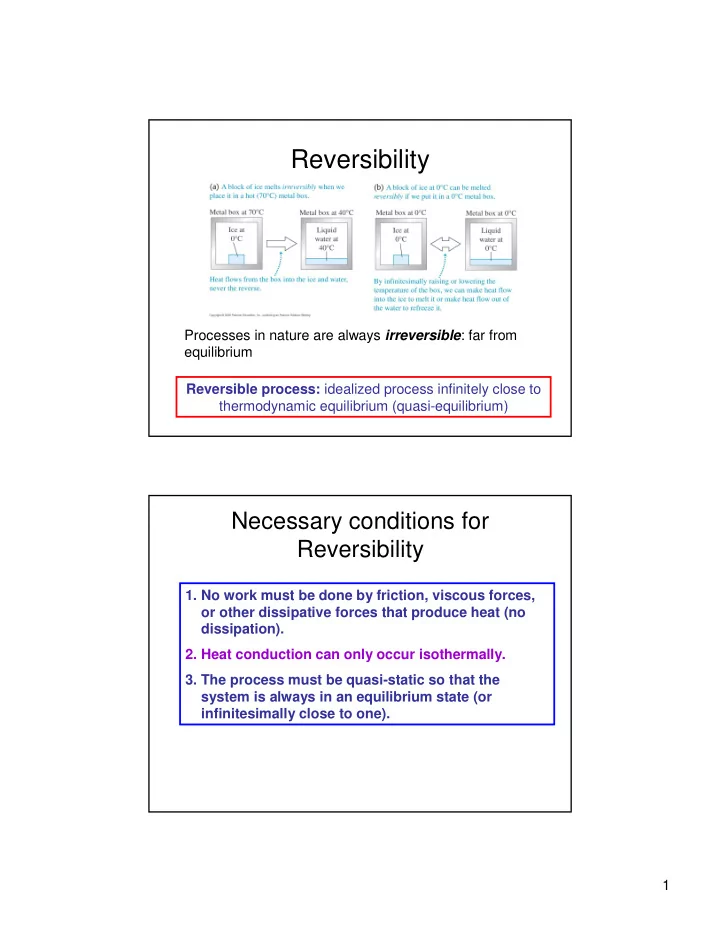
SLIDE 8 8
Entropy change for a Carnot cycle
C C C H H H
T Q S T Q S | | ; | | = ∆ − = ∆
The entropy change in the hot and cold reservoirs is: We know that for a Carnot cycle:
H C H C
T T Q Q = | | | |
) / ( | | | | | | | | = + − = + − = ∆
H C H H H C C H H Total
T T T Q T Q T Q T Q S
The entropy change during a reversible cycle is zero: the entropy change from a state 1 to a state 2 is independent of the path: S is a state function
Entropy of an ideal gas
V dV nRT dT C dQ pdV dU dW dU dQ
V
+ = + = + =
For an ideal gas: This Eq. cannot be integrated unless we know how V depends on T. This another way of saying that Q is not a state function. However, if we divide by T:
dS V dV nR T dT C T dQ
V
= + =
If we assume that Cv remains constant, we can readily integrate it:
1 2 1 2
ln ln V V nR T T C T dQ S
V
+ = = ∆
∫