1
v P
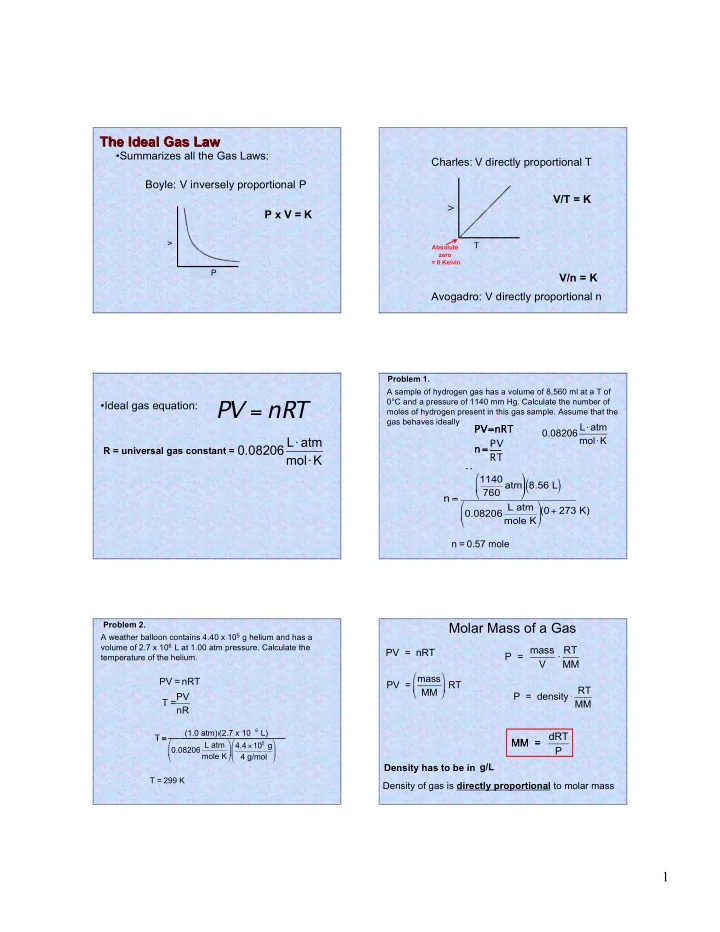
The Ideal Gas Law The Ideal Gas Law
- Summarizes all the Gas Laws:
Boyle: V inversely proportional P P x V = K
V T
Absolute zero = 0 Kelvin
Charles: Avogadro: V directly proportional T V directly proportional n V/T = K V/n = K
PV = nRT
- Ideal gas equation:
R = universal gas constant =
0.08206 L atm molK
Problem 1. A sample of hydrogen gas has a volume of 8,560 ml at a T of 0°C and a pressure of 1140 mm Hg. Calculate the number of moles of hydrogen present in this gas sample. Assume that the gas behaves ideally
PV=nRT PV= n = n = 0.57 mole n = PV=nRT n = PV RT RT 1140 760 atm
- 8.56 L
( )
=
- 0.08206 L atm
mole K
- (0 + 273 K)
0.08206 L atm molK
Problem 2. A weather balloon contains 4.40 x 105 g helium and has a volume of 2.7 x 106 L at 1.00 atm pressure. Calculate the temperature of the helium.
PV = nRT T =
T = T = 299 K T =
- (1.0 atm)(2.7 x 10
(1.0 atm)(2.7 x 10
6 L)
= 0.08206 L atm mole K
- 4.4 105 g
4 g/mol
- = PV
nR
Molar Mass of a Gas
molar mass PV = nRT PV = Density of gas is directly proportional to molar mass MM = P = P = mass V
- mass RT
MM density RT MM MM = dRT P = mass MM
- RT
- RT
Density has to be in g/L