pV=nRT How much does all the air in the room weigh? How much does - - PowerPoint PPT Presentation
pV=nRT How much does all the air in the room weigh? How much does - - PowerPoint PPT Presentation
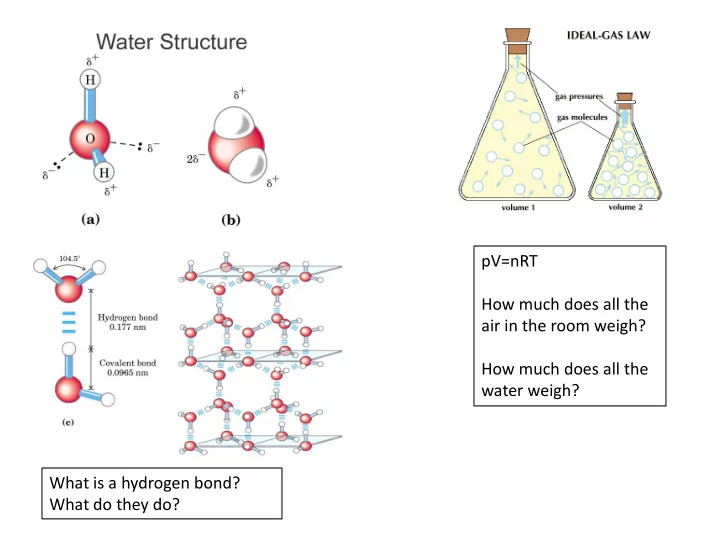
pV=nRT How much does all the air in the room weigh? How much does all the water weigh? What is a hydrogen bond? What do they do? What is an ice crystal? What is a snowflake? A collection of organized water A collection of
What is an ice crystal?
- A collection of organized water
molecules, below 0º C
- Crystal lattice of 4 nearest neighbors,
anchored by H-bonds
- In the atmosphere, grown from vapor,
- ften but not always by Bergeron-
Findeisen process
- Crystallographic symmetry becomes
expressed macroscopically
- Initiated by several ice nucleation
mechanisms
What is a snowflake?
- A collection of individual ice
crystals falling as a hydrometeor
- Often consists of 5-100 ice
crystals
- Size and crystal numbers depend
- n temperature, crystal habits,
and turbulence
- Snowflakes are not made with
scissors
- Density of ice 1h near 0 C: 917kg/m3
- Heat Capacity: 1960 J/kg*K
- Latent Heat of sublimation:
- 2.83 x 106 J/kg (to break H-bonds)
- Ice crystal size: several μm to ~5 mm
- Fall speed: to 80 cm/s
- Fastest Growth rate: -14.7 C
Physical Properties of Ice
d=2.76 A basal face 1022 units/cm3 a axes c axis a c
Unit cells of Ice 1h projection view
Experimental Features
- Quadrupole levitation for wall-less environment
- Alternate support on fine glass fiber
- Vertical flow between 0 and 75 cm/s
- Temperature between room and -65 ºC
- Total pressure between atmospheric and 50 hPa
- Supersaturation controlled between 0 and greater than water saturation
Where In The World ?
- Very high specific heat capacity
- High thermal conductivity for a liquid
- Neutral pH
- Exists on earth in 3 phases
- Ice is less dense than liquid
- Nearly universal chemical solvent
- High surface tension
- Large latent heats (energy of phase change)
- Needed as ingredient for beer
Unique physical properties of water:
- For enthalpy to increase,
heat must be absorbed from surroundings. When this energy is removed from the surroundings, it can be used to break hydrogen bonds. This causes the phase change and cools the surroundings due to heat loss.
- When enthalpy decreases,
heat is released to the surroundings due to the formation of new hydrogen bonds. This warms the surroundings
- The water substance has
constant temperature during a phase change
- In many instances,
multiple phase changes are
- ccurring simultaneously
at one surface.
- Temperature, pressure,
and humidity conditions determine which microscopic process dominates
- The macroscopic behavior
is the net result of all
- processes. We often are
concerned only with explaining this net result.
Phase Diagram for Water: Thermodynamic equilibrium
Clausius-Clapeyron Curve
T T R L e e
v v
1 1 exp
T T R L e e
v v
1 1 exp
- What is the difference between actual and saturation vapor
pressure?
- What is steam?
- What conditions must be met to allow boiling to occur?
- How will the boiling temperature change at high altitude?
Thermodynamics of phase changes in water:
- (blue = heat released to surroundings) – condensational warming
- (red = heat added (removed) from surroundings) – evaporative cooling
- Vaporization/Condensation:
Lv=2.27x106 J/kg
- Freezing/Melting:
Lf=3.3x105 J/kg
- “Latent Heat” is a measure of enthalpy change associated with hydrogen
bonds forming or breaking during a phase change.
- Expressions for heating
- How long must I microwave the frozen milk?
- Clouds are classified mainly by their visual characteristics and height
- They look different because they have different contents
- 3 primary types and many sub-types
Stratus Cumulus Cirrus
Stratus Clouds
Characteristics:
- Can be at any altitude – stratus just means that they form a horizontal layer
- They are often at low altitude in bad weather (nimbostratus)
- Fog is a stratus cloud hugging the ground
- They are formed by weak, but widespread vertical motion (~10 cm/s)
- The are made of a moderate density of cloud drops , LWC~.1 g/m3
- Cumulus or cirrus can also form a layer (Stratocumulus and cirrostratus)
Cumulus Clouds
Characteristics:
- Can be at any altitude – cumulus means “heaping”
- They develop more vertically than horizontally.
- When they form rain they become cumulonimbus
- They are formed by strong vertical motion, sometimes 25 m/s updrafts
- Strong vertical motion and cumulus clouds result from free convection
that comes from instability
- If that vertical motion is deep enough, ice can form in upper part of the cloud
- Ice crystals and strong motion -> charge separation ->lightning
- They have the greatest LWC: from .5 to 4 g/m3 depending of updraft rate
Cirrus Clouds
Characteristics:
- Are composed of tiny ice crystals, not liquid cloud drops
- Usually form only when T< -25 C
- They are formed by weak vertical motion (~5 cm/s)
- The are made of a small density of ice crystals , IWC~.05 g/m3
- Sometimes generated by jet exhaust (contrail)
- Often initiated as anvils of cumulus clouds striking the tropopause-lid
- Important effects due to widespread radiative impact
Cloud Height
Cloud height Cloud types
Low (below 2 km, 6500 ft) Fog Stratus Nimbostratus Stratocumulus Stratus fractus Cumulus humulis Mammatus Funnel Middle(2-6 km, 6500-20000ft) Cumulus humulis Cumulus mediocris Stratocumulus Altostratus Altocumulus High (6+ km, 20000 ft+ ) Cirrus Cirrostratus Cirrus uncinus/fibratus Pileus cloud Large vertical span Cumulus castellanus Cumulus congestus Cumulonimbus
http://apollo.lsc.vsc.edu/classes/met130/notes /chapter5/graphics/drop_cloud_ccn.gif CCN: Cloud Condensation Nuclei
- Needed to turn supersaturation into liquid drops (a site is needed for condensation)
- This is referred to as “drop nucleation” – a big uncertainty in the science of clouds
- CCN are preferentially hydrophillic
- Can be composed of dust, bacteria, pollen, pollutants, acid drops, salt, and others
- Ice nuclei have slightly different characteristics
m =
“net” molecules added molecules impinging - molecules vaporizing
Will make it in Made it out Will be rejected
m =
+
- Cloud Drop
~20 um radius
Vapor deposition into water drops
- Average coefficients help determine net mass growth rate
- Relative local coefficients determine habit type
- Local coefficient is a function of temperature and moisture density
- Mechanism for coefficient temperature function is an enduring mystery
- Mechanism of incorporating incident molecule into lattice is also unknown
+
- m=
On the way out Cleared for landing (condensation) Taking off (sublimation) Brief layover Taxiing to lattice (surface diffusion) Actively growing terrace
2D Nucleation
Vapor deposition onto ice surfaces
Bergeron-Findeison Process
- The saturation vapor over water is greater than ice (see phase diagram)
- This is caused by the greater difficulty in breaking 100% vs 80% of H-bonds
- Vapor tries to move from high concentration -> low concentration (law of diffusion)
- Thus, when water and ice surfaces are nearby, the vapor moves from high concentration
(water surface)->lower concentration (ice surface), allowing ice to grow as water evaporates
- This is the major form of ice crystal growth in mixed-phase clouds
- This process contributes to many stages of the precip. process
Little drops->Big drops: Collision-Coalescence
Riming->graupel->Hail
Frozen precip. scenarios
Crystals photographed in cirrus clouds by aircraft-borne probe
Witches concoct a brew to summon a hailstorm.