SLIDE 1
Product distribution studies of the radical chlorination of - - PowerPoint PPT Presentation
Product distribution studies of the radical chlorination of - - PowerPoint PPT Presentation
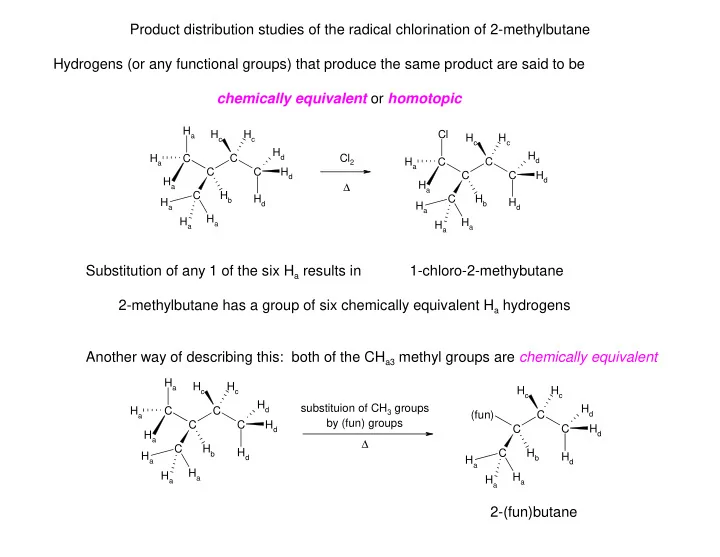
Product distribution studies of the radical chlorination of 2-methylbutane Hydrogens (or any functional groups) that produce the same product are said to be chemically equivalent or homotopic H a H c H c Cl H c H c H d H d H a C C Cl 2 H a C C
SLIDE 2
SLIDE 3
C C C C C Hb Hc Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Hc Hc Hd Hd Hd Cl Ha Ha Ha Ha Ha C C C C C Cl Hc Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha Cl2 ∆
Substitution of the Hb results in 2-chloro-2-methylbutane 2-methylbutane has a group of one chemically equivalent Hb hydrogen The Ha’s and Hb are not chemically equivalent Chemically nonequivalent functional groups are heterotopic functional groups 1-chloro-2-methylbutane
SLIDE 4
C C C C C Hb Hc Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Hc Hc Hd Hd Hd Cl Ha Ha Ha Ha Ha C C C C C Cl Hc Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Cl Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Hc Cl Hd Hd Hd Ha Ha Ha Ha Ha Ha Cl2 ∆
Substitution of an Hc results in (R)-2-chloro-3-methylbutane or (S)-2-chloro-3-methylbutane (a racemate) 2-methylbutane has a group of two chemically equivalent Hc hydrogens The Hc’s generate stereoisomers upon substititution These stereoisomers are enantiomers Functional groups that make stereoisomeric products are stereotopic groups If these stereoisomers are enantiomers, the functional groups are enantiotopic 1-chloro-2-methylbutane 2-chloro-2-methylbutane
SLIDE 5
C C C C C Hb Hc Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Hc Hc Hd Hd Hd Cl Ha Ha Ha Ha Ha C C C C C Cl Hc Hc Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Hc Cl Hd Hd Hd Ha Ha Ha Ha Ha Ha C C C C C Hb Hc Hc Cl Hd Hd Ha Ha Ha Ha Ha Ha Cl2 ∆
Substitution of an Hd results in 1-chloro-3-methylbutane 2-methylbutane has a group of three chemically equivalent Hd hydrogens The CHa3 methyl groups and the CHd3 methyl group are not chemically equivalent So, 2-methylbutane has four groups of chemically nonequivalent hydrogens 1-chloro-2-methylbutane 2-chloro-2-methylbutane rac-2-chloro-3-methylbutane
SLIDE 6
C C C C Hb Hc Hc Hd Hd Hd Ha Ha Ha F C C C C Hb Cl Hc Hd Hd Hd Ha Ha Ha F C C C C Hb Hc Cl Hd Hd Hd Ha Ha Ha F Cl2 ∆
What if a substitution resulted in diastereoisomeric products? (S)-2-fluorobutane (2S,3S)-2-chloro-3-fluorobutane (2R,3S)-2-chloro-3-fluorobutane (S)-2-fluorobutane has two chemically nonequivalent Hc hydrogens The Hc’s generate stereoisomers upon substititution These stereoisomers are diasteriomers Functional groups that make stereoisomeric products are stereotopic groups If these stereoisomers are diasteriomers, the functional groups are diastereotopic So, (S)-2-fluorobutane has five chemically nonequivaent groups of hydrogens
SLIDE 7