SLIDE 1
Nomenclature Common: named as if derived from hydrogen halide, HX - - PowerPoint PPT Presentation
Nomenclature Common: named as if derived from hydrogen halide, HX - - PowerPoint PPT Presentation
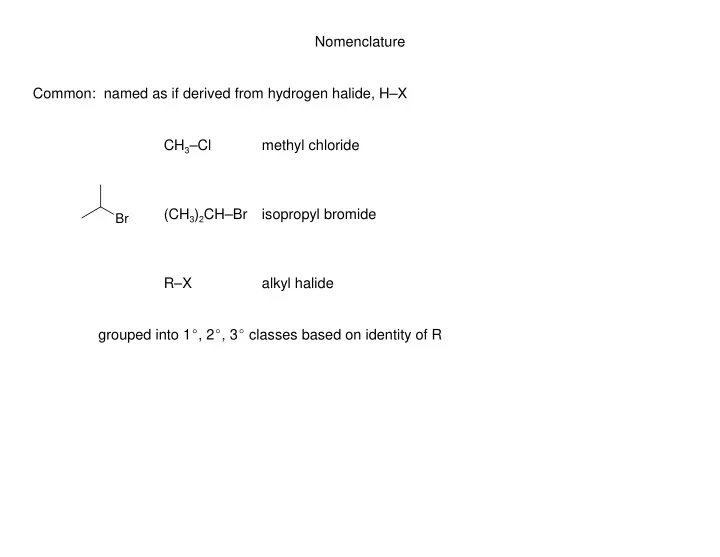
Nomenclature Common: named as if derived from hydrogen halide, HX CH 3 Cl methyl chloride (CH 3 ) 2 CHBr isopropyl bromide Br RX alkyl halide grouped into 1 E , 2 E , 3 E classes based on identity of R Nomenclature IUPAC: named
SLIDE 2
SLIDE 3
Some essential properties of haloalkanes
- R–X are polar
- the C of C–X is intrinsically electrophilic. This dominates the chemistry of this functional group
- R–X have bp’s higher than corresponding R–H
bp - 0.5 EC bp 79 EC
- R–X are toxic
- R–X are relatively rare as natural products
C X H H H δ+ δ−
Cl
SLIDE 4
Cl Cl Cl Cl Cl OCH3 Cl Cl Cl O H CH3 O O Cl Cl OSO3 Cl OH Cl Cl Cl Cl OH Cl OH Cl Cl Cl OH OH
- Synthetic and Naturally Occurring Haloalkanes
DDT (insecticide) methyl ether of a trichloroorcinol LD50 = 115 mg/kg (fungicide produced by water lillies) (compare LD50 acetaminophen = 338 mg/kg) cytotoxic sulfolipid from Mytilus galloprovincialis IC50 = 13 µM
SLIDE 5
O O OH OH Cl O Cl O H HO OH Cl
O O OH OH OH O O H HO OH OH O H
sucralose (Splenda™) 400-800 times as sweet as sugar sucrose
SLIDE 6
Are all nucleophiles created equal? Consider the SN2 reaction of various nucleophiles in a polar protic solvent, holding R–X constant: Nu- + R–X 6 Nu–R + X- Relative Nucleophilicity Nucleophile very good I- HS-, RS- good Br- HO-, RO- N/C- N3- fair Cl-, F- RCO2- NH3 poor H2O, ROH very poor RCO2H
SLIDE 7
Trends in nucleophilicity: BASICITY Consider the SN2 reaction of various nucleophiles in a polar protic solvent, holding R–X constant: Nu- + R–X 6 Nu–R + X- Relative Nucleophilicity Nucleophile very good I- HS-, RS- good Br- HO-, RO- N/C- N3- fair Cl-, F- RCO2-
NH3
poor
H2O, ROH
very poor RCO2H in the same row of the periodic table, stronger bases are better nucleophiles
SLIDE 8
Trends in nucleophilicity: BASICITY Consider the SN2 reaction of various nucleophiles in a polar protic solvent, holding R–X constant: Nu- + R–X 6 Nu–R + X- Relative Nucleophilicity Nucleophile very good I- HS-, RS- good Br- HO-, RO- N/C- N3- fair Cl-, F- RCO2- NH3 poor H2O, ROH very poor RCO2H in the same row of the periodic table, stronger bases are better nucleophiles
SLIDE 9
Trends in nucleophilicity: BASICITY Consider the SN2 reaction of various nucleophiles in a polar protic solvent, holding R–X constant: Nu- + R–X 6 Nu–R + X- Relative Nucleophilicity Nucleophile very good I- HS-, RS- good Br- HO-, RO- N/C- N3- fair Cl-, F-
RCO2-
NH3 poor H2O, ROH very poor RCO2H in the same row of the periodic table, stronger bases are better nucleophiles
SLIDE 10
Trends in nucleophilicity: SIZE AND SOLVATION Consider the SN2 reaction of various nucleophiles in a polar protic solvent, holding R–X constant: Nu- + R–X 6 Nu–R + X- Relative Nucleophilicity Nucleophile very good
I-
HS-, RS- good
Br-
HO-, RO- N/C- N3- fair
Cl-, F-
RCO2- NH3 poor H2O, ROH very poor RCO2H in the same column of the periodic table, bigger is better
SLIDE 11
Trends in nucleophilicity: SIZE AND SOLVATION Consider the SN2 reaction of various nucleophiles in a polar protic solvent, holding R–X constant: Nu- + R–X 6 Nu–R + X- Relative Nucleophilicity Nucleophile very good I- HS-, RS- good Br- HO-, RO- N/C- N3- fair Cl-, F- RCO2- NH3 poor H2O, ROH very poor RCO2H in the same column of the periodic table, bigger is better
SLIDE 12
H O H H O H N H H H O O . . .. . . .. .. . . ..
CH3 C H3 O C H3 N CH3 O H S CH3 C H3 O O O
Why might this be so? SOLVATION MATTERS Polar protic solvents water alcohols ammonia, carboxylic acids amines Polar aprotic solvents ethers acetone DMF DMSO
SLIDE 13
HO- + CH3Br HOCH3 + Br- Ea
relative desolvation in polar aprotic solvents increases the E of the nucleophile: given about the same pKa, smaller is better relative solvation in polar protic solvents decreases the E of the nucleophile: little nucleophiles are more strongly solvated, so given the same pKa, bigger is better
SLIDE 14
H H H H H H H H H H H H H H H H H
Nu-
S S S S S S S S S δ+ δ− E
Nu-
S S S S S S S S
SLIDE 15
H H H H H H H H H
Nu-
S S S S S δ+ δ− E
Nu-
S S S S
SLIDE 16
(CH3)3COCH3 H CH3OH + (CH3)3CBr Ea CH3OH + (CH3)3C+ + Br- + + Br- (CH3)3COCH3 + HBr
While SN2 is favored in polar aprotic solvents, SN1 is favored in polar protic solvents. This is because the polar transition state is stabilized by polar protic solvents, lowering the Ea of the rate limiting step:
SLIDE 17
S O O O H S O O O CF3
- O
O
- O
H O O R R' H + O H R' H + O R H H + S O O O CF3 R S O O O CF3 H S O O O R S O O O
- O
H H H + O R O
Leaving group ability is a function of the basicity of the leaving group
Compound Corresponding acid pKa Corresponding conjugate base
- 11
(R–OTf) (H–OTf) (-OTf) R–I H-I
- 11
I- R–Br H–Br
- 9
Br- R–Cl H–Cl
- 7
Cl-
- 3
(R–OTs) (H–OTs) (-OTs)
- 2
H2O
- 1.5
ROH 5 R–OH H–OH 16 HO- R–OR’ H–OR’ 16 - 18 RO- R–NH2 H–NH2 36 NH2
SLIDE 18
Br O H O H O H O H O H O H O H O H O H O H O H O H + + + + . . .. .. .. .. . . . . .. .. .. .. .. .. .. .. .. .. . . + RLS PDS + +
SLIDE 19
X H R R R R H R R R R Z + Z- + X- SN
The substitution continuum
What factors influence the balance between SN1 and SN2 for a given class of haloakanes?
SLIDE 20
H X H H H O- Na+ OH H H O H H
Primary haloalkanes:
- SN2 for all Z-
- Increased steric hindrance in ‡ decreases the rate
P α-branching in haloalkane P bulky nucleophiles
SLIDE 21
Br CN K+ -CN DMSO
Secondary haloalkanes:
- SN2 for all Z- less basic than HO-
- Increased steric hindrance in ‡ decreases the rate of SN2
P α-branching in haloalkane P bulky nucleophiles
- SN1 Z: is neutral and the solvent
P expect rearrangement CA of Nu: pKa H-CN 9.2 H-OH 15.7 H-CH2CH3 50
SLIDE 22
X O O-Na+ O OH O O O O +
Tertiary haloalkanes:
- Don’t see SN2
- SN1 for most Z: and the anions of strong CA