HYDROGEN IN METALS / METALHYDRIDES Andreas Zttel CONTENTS 1) - - PowerPoint PPT Presentation
HYDROGEN IN METALS / METALHYDRIDES Andreas Zttel CONTENTS 1) - - PowerPoint PPT Presentation
HYDROGEN IN METALS / METALHYDRIDES Andreas Zttel CONTENTS 1) Hydrogen interaction with surfaces 2) Hydrides 3) Stability and hydrogen density 4) Complex hydrides HYDROGEN DENSITY 200 Hydrogen density [kg/m 3 ] 150 NH 3 100 carbon liq.
HYDROGEN DENSITY
50 100 150 200 10 20 30 Hydrogen density [kg/m3] Hydrogen density [kg H2/ 100kg storage material]
Andreas Züttel, Switzerland, 17.07.14
- liq. hydro-
carbons comp. H2 gas
- liq. H2
- liq. natural
gas
NH3
carbon hydrates
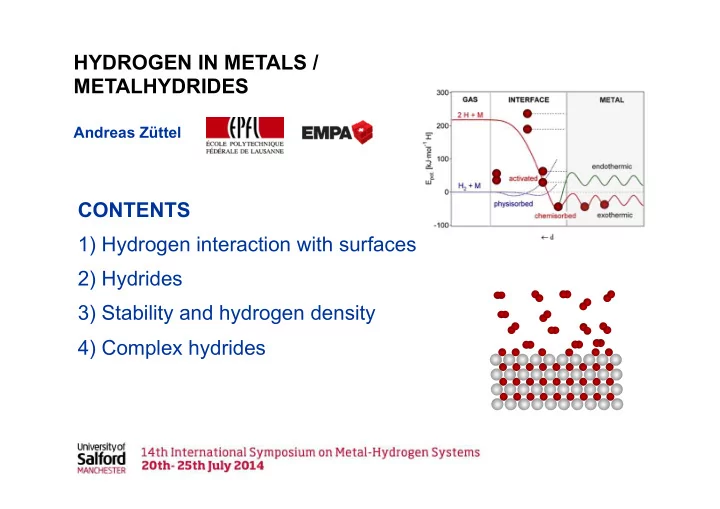
INTERACTION OF GASES WITH SURFACES
S T H G Δ ⋅ − Δ = Δ
Ref: W. Göpel, B. Kühnemann, Z. Phys. Chem. N.F. 122 (1980), p. 75
CHEMISORPTION OF GASES ON METAL SURFACES
Ref: M. Henzler, W. Göpel, „Oberflächenphysik des Festkörpers, Teubner Studienbücher Physik, B. G. Teubner Stuttgart 1991, p. 474
ΔEchem≈ 0.25 eV
LENNARD-JONES POTENTIAL
Ref.: J. E. Lennard-Jones, Trans. Faraday Soc. 28 (1932), pp. 333.
- L. Schlapbach, Chapter 1, L. Schlapbach (Ed.) in Intermetallic Compounds I, Springer Series Topics in Applied
Physics, Vol. 63, Springer–Verlag, 1988, p. 10.
Andreas Züttel, Switzerland, 17.07.14 6
BINARY HYDRIDES
Ref.: Gottfried Brendel, Kapitel: Hydride, Ullmanns Encyklopädie der technischen Chemie, 4. neubearbeitete und erweiterte Auflage, Band 13 (1977), pp. 109-133, Verlag Chemie Weinheim New York
DISCOVERY OF HYDROGEN ABSORPTION IN METALS
Thomas GRAHAM, born Dec. 20, 1805, Glasgow, Scot., died
- Sept. 11, 1869, London, Eng.
British chemist often referred to as the father of colloid chemistry. Educated in Scotland, Graham persisted in becoming a chemist, though his fat- her disap- proved and with- drew his support. He then made his living by writing and teaching. He was a professor at a school in Edinburgh (1830–37) and at University College, London (1837– 55), and was master of the mint (1855–69). In his final paper he described palladium hydride, the first known instance of a solid compound formed from a metal and a gas.
Ref.: Thomas Graham„On the Occlusion of Hydrogen Gas by Metals“, Proc. Royal Soc. 16 (1868), pp. 422
8
HYDROGEN ABSORPTION IN METALS
- Ph. Mauron, M. Bielmann, EMPA, Switzerland
9
HYDROGEN ABSORPTION IN METALS
0 sec (vacuum) 5 sec (8 bar H2) 8 sec (8 bar H2) 15 sec (8 bar H2) 20 sec (8 bar H2) 30 sec (7 bar H2) 45 sec (6 bar H2) 60 sec (5 bar H2) ZrMn1.5
HYDROGEN IN AND ON PALLADIUM
THERMO DESORPTION SPECTROSCOPY After exposing Pd(111) at 115 K to equal doses of H2 and D2 a much stronger desorption of α-H than α-D is observed apparently due to faster H than D absorption into α-states. Only minor HD desorption was ob-
- served. Apparently the gas which was
dosed second bypassed the chemi- sorption state. α: chemisorbed hydrogen at the surface β: interstitial hydrogen
Ref.: G. E. Godowski, R. H. Stulen, T. E. Felter, J.
- Vac. Sci. Technology A5 (1987), pp. 1103
α β
HYDROGEN IN AND ON PALLADIUM
Andreas Züttel, IfRES (2005) 12
THERMODYNAMICS OF HYDRIDES
ΔH0 = 2/n·ΔHf M + n/2 H2 MHn
H2
ΔHf M + n/2 H2 → MHn
( ) ( ) ( )!
" # $ % & Δ − Δ − Δ ⋅ = Δ
2
2 2 H H n M H MH H n H
n
( ) ( ) ( )!
" # $ % & − − ⋅ = Δ
2
2 2 H S n M S MH S n S
n
S0(MHn) ≈ S0(M) → ΔS0 ≈ -S0(H2) S0(H2) = 130 JK-1mol-1 ΔH
= 0 = 0 ≈ 0
HYDROGEN IN METALS
Ref.: P. Nordlander, J. K. Norskov, and F. Besenbacher, J. Phys. F. Metal Phys. 16:9 (1986), pp. 1161-1171, J.K. Norskov and F. Besenbacher, J. of the Less-Common Metals 130 (1987), pp. 475-490
METAL H n r
EFFECTIVE MEDIUM THEORY
Jens Norskov
Andreas Züttel, Switzerland, 17.07.14 1 4
HEAT OF SOLUTION
LATTICE GAS
The free energy F = U - T·S is
ln exp
H
- HH
N N F kT kT ε ε + " # = − − % & ' (
∑
N : total number of sites NH : number of H ε0 : H binding energy NHH : number of nearest neighbour H-pairs ε : H-H pair interaction energy
- Ref. Ronald Griessen, VU Amsterdam, NL
Hemmes H, Salomons E, Griessen R, Sänger P, Driessen A., Phys Rev B Condens Matter. (1989);39(15):10606-10613.
ε ε
HH
- H
N N E + =
ε ε0
ISOTHERM
- Ref. Ronald Griessen, VU Amsterdam, NL
ln ) ( ln 2 1 → → ⋅ ⋅ ≅ ⋅ ⋅ ⋅
H H
c p c T k T p p T k
) ( 1 ln
2 1 =
− ⋅ ⋅ + − ⋅ ⋅
i i i
c n c c T k ε
b dis
n T p p T k ε ε ε − ⋅ + ⋅ = ⋅ ⋅ 2 ) ( ln
H H b H
c c k T c n T p p T k − ⋅ ⋅ + − ⋅ ⋅ + = ⋅ ⋅ ⋅ 1 ln ) ( ln
2 1 2 1
ε ε ε
ΔH ΔS ΔG Solubility (Sieverts) Plateau (Maxwell) Coexistence curve
Ronald Griessen
Andreas Züttel, Switzerland, 17.07.14 1 7
HYDROGEN ABSORPTION IN METALS α-Phase: Solid Solution MHx (0 < x < 0.1) H H, ΔV/V = k·cH β-Phase: Hydride Phase MHx x = {1, 2, 3,…} H H
R H Δ −
R S Δ
R S T R H p p Δ + ⋅ Δ − = $ $ % & ' ' ( ) ln
( )
H
c T k p p ln ln ⋅ ⋅ = " " # $ % % & ' 2 1
( )
H
c T k p p ln ln ⋅ ⋅ = " " # $ % % & ' 2 1
STABILITY OF HYDRIDES: VAN’T HOFF PLOT Tdec
Andreas Züttel, IfRES (2005) 19
THERMODYNAMICS OF HYDRIDES
ΔH0 = 2/n·(ΔHf,H - ΔHf,M) x A + y B + n/2 H2
H2
ΔHf,M x A + y B + n/2 H2 → AxBy + n/2 H2 → AxByHn
( ) ( )
( )!
" # $ % & Δ − Δ − Δ ⋅ = Δ
2
2 2 H H n B A H H B A H n H
y x n y x
( ) ( )
( )!
" # $ % & − − ⋅ = Δ
2
2 2 H S n B A S H B A S n S
y x n y x
S0(AxByHn) ≥ S0(AxBy) → (ΔS0( ≤(-S0(H2)( S0(H2) = 130 JK-1mol-1 AxByHn AxBy + n/2 H2 ΔHf,H ΔH
Andreas Züttel, Switzerland, 17.07.14 2
ELECTRONIC STRUCTURE
1) Lattice expansion reduces the band- width. 2) Attractive potential of the proton affects the metal states and leads to the metal- hydrogen band 5 to 8 eV below EF. 3) The H-H interaction results in new features in the lower part of the density of states for systems with more than one H atom per unit cell. 4) An upwards shift of EF results from the balance between the additional electrons brought in by hydrogen and the number of new states below EF. The balance between the ‘exothermic’ lowering of occupied states and the ‘endothermic’ upwards shift of EF d e t e r m i n e s t h e s t a b i l i t y o f t h e metalhydride. Ref.: L. Schlapbach, F. Meli, and A. Züttel, Chap. 21: “Intermetallic Hydrides and their Applications” in Intermetallic Compounds: Vol. 2, Practice, J. H. Westbrook and R. L. Fleischer (1994) John Wiley & Sons Ltd.
Bandstructure of LaNi5 and LaNi5H7
BINDING ENERGY
Ref.: Hammer B; Norskov J K, “Why Gold is the Noblest of all the Metals”, Nature 376, (1995), pp. 238-240
Energy antibonding bonding EF noble metal EF transition metal Adsorbate level
H s s, p d EF DoS E
Andreas Züttel, Switzerland, 17.07.14 2 2
SEMI-EMPIRICAL MODEL FOR THE STABILITY
The Local Band-Structure Model a = 18.6 kJ·mol-1HÅ4eV-3/2 b = -90 kJ·mol-1H
b R W E a H
j 4 j
+ ∑ ⋅ ⋅ Δ ⋅ = Δ
− ∞ Rj
Ref.: R. Griessen, Phys. Rev. B 38 (1988), pp.3690-3698 and V.L. Moruzzi, J. F. Janak, A.R. Williams, „Calculated Electronic Properties of Metals, Pergamon, New York (1978) ΔE W
EMPIRICAL MODELS: STABILITY
1) Reversed stability (global)
ΔH(ABnH2m) = ΔH(AHm) + ΔH(BnHm) - (1-F)·ΔH(ABn)
Miedema Model
Ref.: H.H. Van Mal, K.H.J. Buschow and A.R. Miedema, J. Less-Common Met. 35 (1974), pp. 65
2) Imaginary binary hydrides (local) AmBn + xH → (AmBnHx) interstitial site
ΔH([AaBb]H) = ΔH(AmHx·a/(a+b)) + ΔH(BnHx·b/(a+b))
binary hydrides
Ref.: I. Jacob, J.M. Bloch, D. Shaltiel and D. Davidov, Solid State Comm. 35 (1980),
- pp. 155.
Andreas Züttel, Switzerland, 17.07.14 2 4
INTERSTITIAL SITES IN METAL HYDRIDES HYDROGEN ON TETRAHEDRAL SITES HYDROGEN ON OCTAHEDRAL SITES
Ref: J. J. Reilly, G. D. Sandrock, Metallhydride als Wasserstoff-Speicher, Spektrum der Wissenschaften (April 1980), pp. 53-59
Andreas Züttel, University of Fribourg, 15.12.2002 25
FAMILIES OF HYDRIDE FORMING INTERMETALLICS Intermetallic Prototype Structure compound AB5 LaNi5 Haucke phases, hexagonal AB2 ZrV2, ZrMn2, TiMn2 Laves phase, hexagonal or cubic AB3 CeNi3, YFe3 hexagonal, PuNi3-typ A2B7 Y2Ni7, Th2Fe7 hexagonal, Ce2Ni7-typ A6B23 Y6Fe23 cubic, Th6Mn23-typ AB TiFe, ZrNi cubic, CsCl- or CrB-typ A2B Mg2Ni, Ti2Ni cubic, MoSi2- or Ti2Ni-typ
BODY CENTERED CUBIC SOLID SOLUTION ALLOYS
BCC Alloys: Ti-V-Mn, Ti-V-Cr, Ti-V-Cr-Mn, and Ti-Cr-(Mo, Ru)
Ref.: E. Akiba and M. Okada, “Metallic Hydrides III: Body-Centered-Cubic Solid-Solution Alloys”, MRS BULLETIN/SEPTEMBER 2002 699-703
V VH VH2 Structure fcc & hcp bcc Site O T O T Number 1 2 3 6 Size 0.414 0.255 0.155 0.291
DENSITY OF STATES FOR HYDROGEN
Andreas Züttel, Switzerland, 17.07.14 2 8
DENSITY OF STATES FOR HYDROGEN
Ref.: I. Bakonyi, F. Mehner, A. Rapp, A. Cziraki, E. Toth-Kadar, V. Skumryev, R. Reisser, H. Kronmüller and
- R. Kirchheim , Zeitschrift für Metallkunde (1993)
Andreas Züttel, Switzerland, 17.07.14 2 9
EMPIRICAL MODELS: GEOMETRY 1) Size of interstitial site: r > 0.37 Å Westlake criterion
Ref.: D. G. Westlake, J. Less-Common Metals 91 (1983), pp.275-292
2) Distance between hydrogen atoms: d > 2.1 Å
Ref.: A. C. Switendick, Z. Phys. Chem. N.F. 117 (1979), pp. 89
Number of hydrogen atoms:
Number of interstitial sites for which 1) and 2) applies.
ρV < 245 kg m-3
Andreas Züttel, Switzerland, 17.07.14 3
METAL HYDRIDES WITH SHORT H-H SEPARATIONS RTInH1.333 (R = La, Ce, Pr, or Nd; T = Ni, Pd, or Pt)
Ref.: P. Vajeeston et al. Phys. Rev. B 67 (2003), 014101 charge transfer electron density
CATALYZED HYDROGEN ABSORPTION
H2 activated Complex Desorption Recombination Adsorption Intercalation Mobility
- A. Züttel et al., LiBH4 a new
hydrogen storage material, Journal of Power Sources 118 (2003), pp. 1–7
- S. Orimo et al., "Dehydriding
and rehydriding reactions of LiBH4", Journal of Alloys and Compounds 404-406 (2005),
- pp. 427-430
- B. Bogdanovic, M. Schwickardi, Ti-
doped alkali metal aluminium hydrides as potential novel reversible hydrogen storage materials, Journal of Alloys and Compounds 253, 1-9 (1997).
1996 - 1997
- J. N. Huiberts, R. Griessen, J. H.
Rector, R. J. Wijngaarden, J. P. Dekker, D. G. de Groot, N. J. Koeman, Yttrium and lanthanum hydride films with switchable
- ptical properties, Nature 380,
231-234 (21 March 1996)
2003 - 2004
“LiBH2” “LiBH3.6” “LiBH3” LiH
STABILITY OF HYDRIDES
ΔH0 Elements Alloy Hydride Complex ΔHf ΔHdec
Li[BH4] LiH+B Li7B6 Li + B + H2
- 195
[kJ]
- 90
- 75 kJ/molH2
H2
Andreas Züttel, IfRES (2005) 34
THERMODYNAMICS OF HYDRIDES
ΔH0 = 2/(3n)·(ΔHf,C - ΔHf,P) A + n B + 2n H2
H2
ΔHf,P A + n B + 2n H2 → AHn + B + 3/2n H2 → A[BH4]n
[ ] ( ) ( ) ( )!
" # $ % & Δ − Δ − Δ ⋅ = Δ
2 4
2 3 2 H H n AH H BH A H n H
n n
( ) ( )
( )!
" # $ % & − − ⋅ = Δ
2
2 2 H S n B A S H B A S n S
y x n y x
S0(A[BH4]n) < S0(AHn + B) → (ΔS0( > (-S0(H2)( S0(H2) = 130 JK-1mol-1 A[BH4]n AHn + B + 3/2n H2 ΔHf,C ΔH
DEVELOPMENT*OF*THE*HYDROGEN*DENSITY*
5 10 15 20 25 1850 1900 1950 2000 2050 Year H/MH [mass%]
Pd* Mg2NiH4* NaAlH4* LiBH4* NH3BH3*
CH4* liquid*e.g.*Al[BH4]3*
!" +" Thomas"GRAHAM" Shin!Ichi"ORIMO" """""""""""Andreas"ZÜTTEL" Boris"BOGDANOVIC"
Andreas Züttel, IfRES (2005) 36
STABILITY OF BHn AND BHn-
Ref.: Puru Jena , Virginia Commonwealth University, Richmond, VA (to be published).
Gradient Corrected Density Functional Theory Energy gain in adding a H atom BHn-1 + H → BHn
Puru Jena
ADSORPTION ENERGY
[ ] ( )
( )
2 2 1 1
97
H M dis HH dis MM dis MH
X X H H mol kJ H − ⋅ + Δ + Δ ⋅ = ⋅ Δ
−
Pauling: Absorption Energy:
[ ]
( )
2 1
194
H M dis MM ads
X X H mol kJ H − ⋅ + Δ = ⋅ Δ
−
Pauling electronegativity
Ref.: Linus Pauling, “THE PRINCIPLES DETERMINING THE STRUCTURE OF COMPLEX IONIC CRYSTALS“, Journal of the American Chemical Society, 1929 - pubs.acs.org
Linus Carl Pauling
* 28. 2. 1901; † 19. 8. 1994 1954 Nobelprize for chemistry 1962 Nobelprize for peace
Andreas Züttel, IfRES (2005) 38
STABILITY OF COMPLEX HYDRIDES
M[BH4]n M, B, nH2
ΔH
ΔHdis M, B, H2 ΔHm ΔHMH = ΔHMM + 194·(XM - XH)2 [kJ / mol H] MHn + Bn + 3/2nH2 M[BH4]n ΔHf
0 = 247.4·XM - 421.2
[kJ / mol BH4] ΔHdis = ΔHf
0 - ΔHMH - ΔHm
Ref.: Y. Nakamori, K. Miwa, A. Ninomiya, H. Li, N. Ohba, S.-I. Towata, A. Züttel, and Shin-ichi Orimo, Physical Review B 74, 045126 (2006); Linus Pauling, THE PRINCIPLES DETERMINING THE STRUCTURE OF COMPLEX IONIC CRYSTALS, Journal of the American Chemical Society, 1929 - pubs.acs.org
Pauling Miwa, Orimo
50 100 150 200 10 20 30 Hydrogen density [kg/m3] Hydrogen density [kg H2/ 100kg storage material]
Andreas Züttel, Switzerland, 17.07.14
HYDROGEN DENSITY
carbon hydrates
- liq. hydro-
carbons metal hydrides comp. H2 gas
- liq. H2
- liq. natural
gas
NH3