Technology Overview Current Status
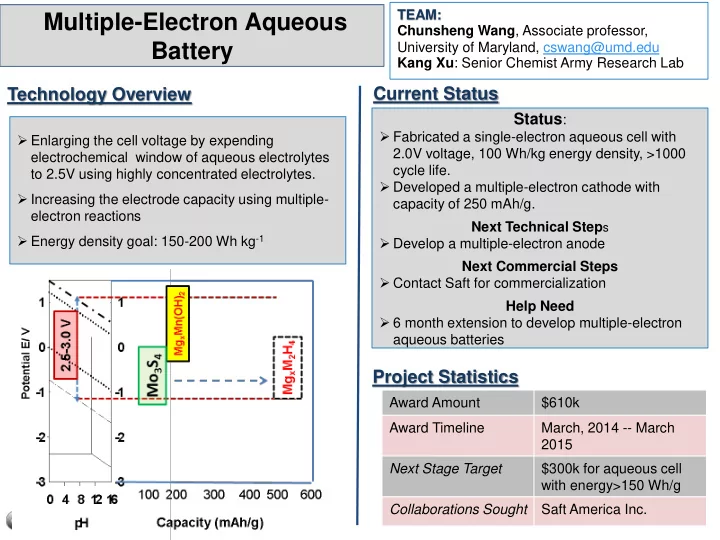
- Enlarging the cell voltage by expending
electrochemical window of aqueous electrolytes to 2.5V using highly concentrated electrolytes.
- Increasing the electrode capacity using multiple-
electron reactions
- Energy density goal: 150-200 Wh kg-1
Status:
- Fabricated a single-electron aqueous cell with
2.0V voltage, 100 Wh/kg energy density, >1000 cycle life.
- Developed a multiple-electron cathode with
capacity of 250 mAh/g. Next Technical Steps
- Develop a multiple-electron anode
Next Commercial Steps
- Contact Saft for commercialization
Help Need
- 6 month extension to develop multiple-electron
aqueous batteries
Multiple-Electron Aqueous Battery
TEAM: Chunsheng Wang, Associate professor, University of Maryland, cswang@umd.edu Kang Xu: Senior Chemist Army Research Lab
Project Statistics
Award Amount $610k Award Timeline March, 2014 -- March 2015 Next Stage Target $300k for aqueous cell with energy>150 Wh/g Collaborations Sought Saft America Inc.