HOT CORROSION HOT CORROSION Metals and alloys experience accelerated - - PowerPoint PPT Presentation
HOT CORROSION HOT CORROSION Metals and alloys experience accelerated - - PowerPoint PPT Presentation
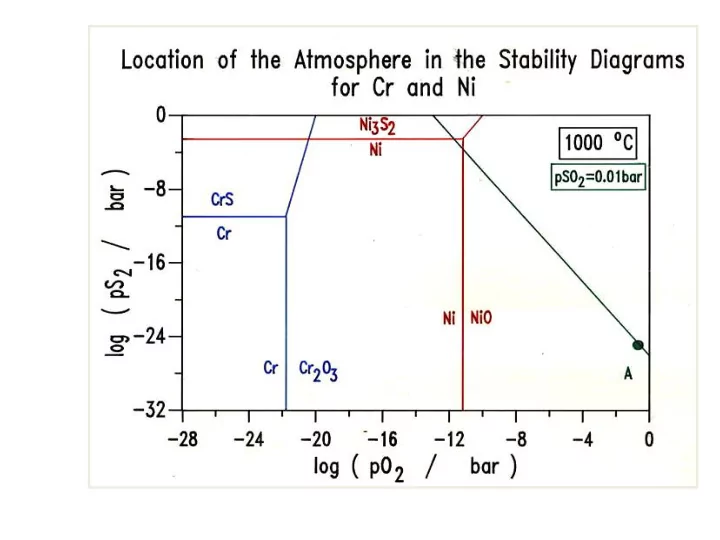
Relationship between pS 2 and pSO 2 HOT CORROSION HOT CORROSION Metals and alloys experience accelerated oxidation / sulphidation when their surfaces are covered with a thin film of fused salt in an oxidizing gas atmosphere at elevated
Relationship between pS2 and pSO2
HOT CORROSION
HOT CORROSION Metals and alloys experience accelerated oxidation / sulphidation when their surfaces are covered with a thin film of fused salt in an oxidizing gas atmosphereat elevated temperatures
- Temp. range 850-950oC (m.p.
- f Na2SO4 884°C)
- Initial attack on protective
- xide film
- Chromium depletion,
- xidation of the base material
accelerates
Type I (HTHC)
- Temp. range 650-800oC
- Formation of low melting
mixtures Na2SO4-NiSO4 eutectics
- High partial pressure of SO3
(g) –required
- Typical pitting
Type II (LTHC)
Types of Hot Corrosion
HIGH TEMPERATURE HOT CORROSION
Ref: Introduction to High Temperature Oxidation and corrosion, A.S.Khanna, IIT Bombay, ASM International, 2002
- Occurs at high temperature
- Salt deposit on metal / alloy in liquid state
- Very high corrosion rate
- Kinetics - mainly linear
- Corrosion of Ni in presence of Na2SO4
at 900 oC
- Melting point of Na2SO4 - 880 oC
LOW TEMPERATURE HOT CORROSION
Ref: Introduction to High Temperature Oxidation and corrosion, A.S.Khanna, IIT Bombay, ASM International, 2002
- Reaction rate at beginning – slow till certain point – incubation period
- Later rate increases suddenly and follow linear kinetics
- Occurs at lower temperatures – lower than melting temperature of salt
- Initial stages - Na2SO4 solid
– corrosion reaction involves
- xidation of Ni to NiO
- NiO + SO2 = NiSO4
- Melting point of NiSO4 - 671
- C
FLUXING MECHANISM OF HOT CORROSION
Oxyanion sodium sulphate melt similar to acid-base chemistry of aqueous solutions Na2O + SO3 = Na2SO4 log K (1200 K) = -16.7 log aNa2O = melt basicity log aSO3 = melt acidity
Ref: Robert A. Rapp, Hot corrosion of materials: a fluxing mechanism? Corrosion Science 44 (2002) 209±221
Dissolution on NiO can be represented by the following equations: Basic dissolution: 2NiO + O2- (Na2O) + ½ O2 = 2NiO2- (NaNiO2) Acidic dissolution: NiO = Ni2+ + O2- Further O2- + SO3 = SO4
2- (NiSO4)
SUSTAINED ACIDIC DISSOLUTION
Basic dissolution 2MO + O2- (Na2O) + ½ O2 = 2MO2- (NaNiO2) melt gradually consumes the salt gets saturated with NaNiO2 reaction subsides Acidic dissolution MO = M2+ + O2- Further O2- + SO3 = SO4
2-
(NiSO4) molten Na2SO4 – solvent for NiSO4 which reacts with nickel migrating
- utwards through the inner layer with a
continuous sulphide network Na2SO4 is not consumed and the reaction continues for extended periods
- f time
Crucible Test Schematic of Bead Test Isothermal Tests Cyclic Tets
Schematic of a Burner Rig
Hot Corrosion of Nickel
FORMATION OF EUTECTIC MIXTURE
Temperature dependence of the corrosion rate of Ni-30% Cr coated with Na2SO4 (2.5 mg/cm2) in latm. of O2 + 1% (SO2 + SO3) and Ni-20% Cr (with no salt deposit) in 1 atm. of SO2 : O2 : 1 : 1.
Na2SO4 induced hot corrosion of Ni-30% Cr and Co-30% Cr in O2 + 1% (SO2 + SO3) and O2 + 0.15% (SO2 + SO3) exposed for 24 h.
ROLE OF ALLOYING ELEMENTS
- Improves mechanical properties
- Presence makes alloy highly susceptible to hot
corrosion Vanadium and molybdenum
- increase the hot corrosion resistance.
Titanium, aluminum, and niobium