SLIDE 1
ASYMMETRIC HYDROGENATION OF KETONES O OH chiral Ru cat H 2 R - - PowerPoint PPT Presentation
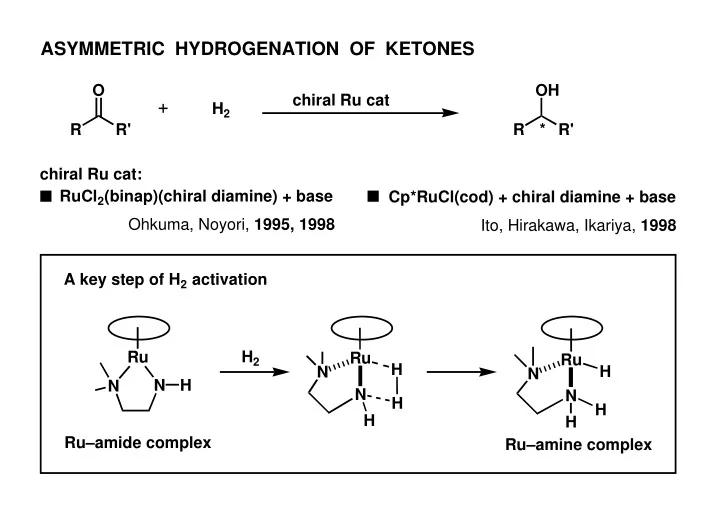
ASYMMETRIC HYDROGENATION OF KETONES O OH chiral Ru cat H 2 R - - PowerPoint PPT Presentation
ASYMMETRIC HYDROGENATION OF KETONES O OH chiral Ru cat H 2 R R' R * R' chiral Ru cat: RuCl 2 (binap)(chiral diamine) + base Cp*RuCl(cod) + chiral diamine + base Ohkuma, Noyori, 1995, 1998 Ito, Hirakawa, Ikariya, 1998 A key
SLIDE 2
SLIDE 3
CATALYST PRECURSOR FOR THE HYDROGENATION
C6H5 O C6H5 OH 2-propanol H2 + Ru cat 10 atm ketone:Ru:diamine = 1000:1:1 (CH3)2N(CH2)2NH2 30 °C, 2 h Ru cat: Ru Ru O O TOF = 100 TOF = product mol/cat mol·h H3C CH3
SLIDE 4
SOLVENT EFFECT ON THE TURNOVER NUMBERS
solvent 2-propanol TONa ethanol 300 200 methanol 70 THF CH2Cl2 DMF 65 70 CH3CN 30 hexane tert-butyl alcohol 20
aTON = product mol/cat mol
Ru cat 0.2 mol%, H2 10 atm, 30 °C, 2 h Conditions: Ru cat = (Cp*RuOCH3)2 + (CH3)2N(CH2)2NH2
SLIDE 5
solvent H2 or D2 + Cp*RuCl(cod) (CH3)2N(CH2)2NH2 KOH 1 atm
ISOTOPE LABELING EXPERIMENTS
30 °C, 2–3 h OD OH D OH H2 D2 D2 7 90 ketone:Ru:diamine:KOH = 100:1:1:1 H2 or D2 solvent D content, %a
a Determined by 1H NMR and D NMR.
~100% yield C6H5 O C6H5 OH H (D)
SLIDE 6
OH C6H5 N Ru H NH C H O C6H5
A POSSIBLE MECHANISM
Ru HN N Ru H H2N N N Ru HN H H N Ru N H H O C6H5 H R O H Cp*RuCl(cod) KOH (Cp*RuOCH3)2 (CH3)2N(CH2)2NH2 (CH3)2N(CH2)2NH2 + + + H2 ROH ROH
SLIDE 7
ASYMMETRIC HYDROGENATION WITH CHIRAL Ru CATALYSTS
C6H5 OH 72% ee (R) C6H5 OH C6H5 OH C6H5 OH C6H5 OH C6H5 OH 79% ee (R) 79% ee (R) 73% ee (R) 88% ee (R) 81% ee (R) C6H5 R O C6H5 R OH 30 °C, 6–18 h H2 + KOH 10 atm ketone:Ru:diamine:KOH = 100:1:1:1 Cp*RuCl(cod) chiral diamine 2-propanol NH2 N C2H5 chiral diamine
S R
examples:
SLIDE 8
EFFECT OF STRUCTURES OF DIAMINES ON THE ee VALUES
NH2 N C2H5 N H NH2 N H N(CH3)2 N H N N H N CH3 3% ee (R) 72% ee (R) 13% ee (S) 40% ee (S) 13% ee (R)
SLIDE 9