SLIDE 1
Cp*Ru(II) COMPLEXES BEARING PRIMARY AMINE LIGANDS L = N(CH 3 ) 2 - - PowerPoint PPT Presentation
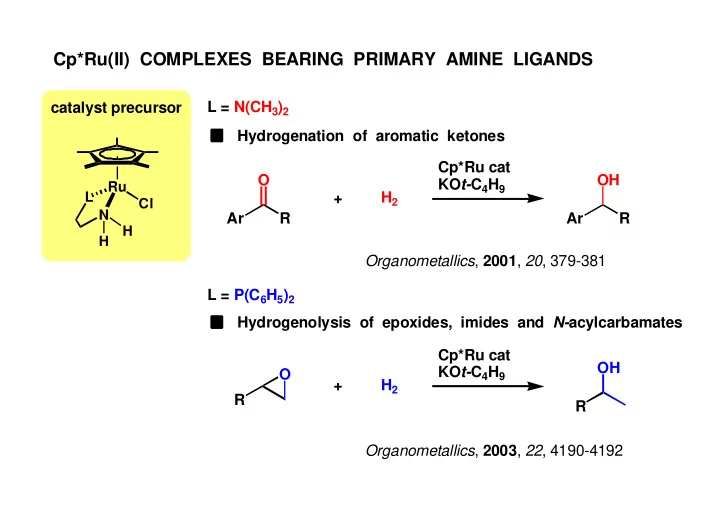
Cp*Ru(II) COMPLEXES BEARING PRIMARY AMINE LIGANDS L = N(CH 3 ) 2 - - PowerPoint PPT Presentation
Cp*Ru(II) COMPLEXES BEARING PRIMARY AMINE LIGANDS L = N(CH 3 ) 2 catalyst precursor Hydrogenation of aromatic ketones Cp*Ru cat O OH KO t- C 4 H 9 Ru L H 2 + Cl N Ar R Ar R H H Organometallics , 2001 , 20 , 379-381 L = P(C 6
SLIDE 2
SLIDE 3
M NCCH3 Ph2 P N H2 (OTf)2
HYDROGENOLYSIS OF STYRENE OXIDE WITH [Cp*M(CH3CN)(Ph2PCH2CH2NH2)](OTf)2
C6H5 O + C6H5 H2 OH O M = Rh, Ir 2-propanol 30 °C, 4 h Rh, Ir cat KOt-C4H9 H2, atm conv, %a 10 10 >99 >99 epoxide:cat:KOt-C4H9 = 100:1:1, [epoxide] = 0.5 M in 2-propanol
adetermined by 1H NMR. b[Cp*Ru(CH3CN)(Ph2PCH2CH2NH2)](OTf). cC6H5CH(CH3)OH:C6H5CH2CH2OH = 78:22.
α:βa catalyst precursor cat Rh/KOt-C4H9 Ir/KOt-C4H9 >99 >99 entry 2 3 4 5 Rh Ir C6H5 O OH + α β 6 KOt-C4H9 <1 93:7 93:7 95:5 91:9 1 Rub/KOt-C4H9 10 >99 hydrogenated productc
SLIDE 4
Ru NCCH3 L N H2 OTf catalyst precursor
HYDROGENOLYSIS OF 1,2-EPOXYOCTANE WITH [Cp*Ru(CH3CN)(L—NH2)]OTf
n-C6H13 O n-C6H13 OH H2 + 2-propanol 30 °C, 4 h cat KOt-C4H9 epoxide:cat:KOt-C4H9 = 100:1:1, [epoxide] = 0.5 M in 2-propanol 10 atm electron density on Ru center Brønsted acidity of coordinated NH2 group PPh2 PPh2 N(CH3)2 N PPh2 PPh2 increase increase NH2 NH2 NH2 NH2 NH2 NH2 <1 <1 71 21 >99 87 L—NH2 conv* *determined by 1H NMR
SLIDE 5
ELECTRONIC PROPERTY OF Cp*Ru, Rh, Ir COMPLEXES BEARING P,N-LIGAND
IR (νC≡O) 2099 cm-1 2071 cm-1 Rh CO Ph2 P N H2 (OTf)2 Ir CO Ph2 P N H2 (OTf)2 1948 cm-1 Ru CO Ph2 P N H2 OTf M NCCH3 Ph2 P N H2 M CO Ph2 P N H2 + CO 1 atm 30 °C, 24 h CH2Cl2 (OTf)2 (OTf)2 M = Rh, Ir
SLIDE 6
SELECTED DATA FOR [Cp*Ru(CO)(L–NH2)]OTf
Ru CO Ph2 P N H2 Ru CO Ph2 P N H2 Ru CO Ph2 P N H2 Ru CO Ph2 P N H2 IR (νC≡O) 1948 cm-1 1951 cm-1 1939 cm-1 1947 cm-1 Ru CO Me2 N N H2 IR (νC≡O) 1931 cm-1 Ru NCCH3 Ph2 P N H2 OTf Ru CO Ph2 P N H2 92% yield OTf + CO 1 atm 30 °C, 10 min CH2Cl2 Ru CO N 1938 cm-1 N H2
SLIDE 7
NCCH3 Ru
PREPARATION OF [Cp*M(CH3CN)(Ph2PCH2CH2NH2)]n+
CH3CN CH3CN Ru NCCH3 Ph2 P N H2 >99% yield OTf OTf NH2 PPh2 30 °C, 1 h + CH2Cl2 M Cl Ph2 P N H2 M NCCH3 Ph2 P N H2 >99% yield + 2 AgOTf 30 °C, 24 h CH3CN Cl (OTf)2 M = Rh, Ir
SLIDE 8