SLIDE 1
HYDROGENATION OF KETONES WITH Cp*Rudiamine CATALYST - - PowerPoint PPT Presentation
HYDROGENATION OF KETONES WITH Cp*Rudiamine CATALYST - - PowerPoint PPT Presentation
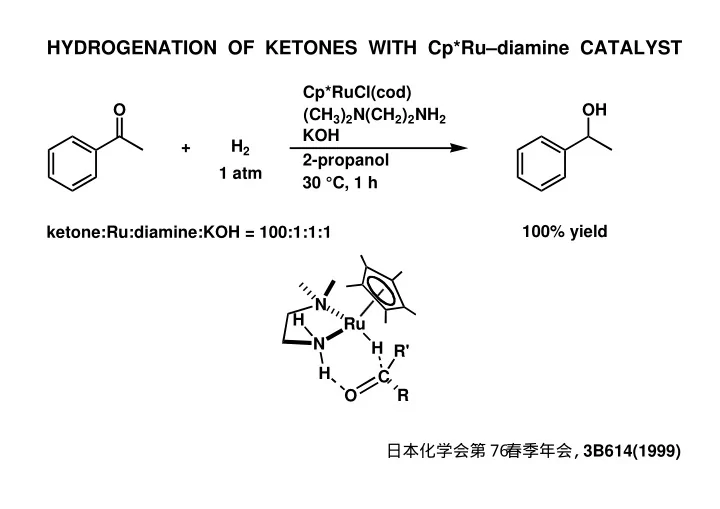
HYDROGENATION OF KETONES WITH Cp*Rudiamine CATALYST Cp*RuCl(cod) O OH (CH 3 ) 2 N(CH 2 ) 2 NH 2 KOH H 2 + 2-propanol 1 atm 30 C, 1 h 100% yield ketone:Ru:diamine:KOH = 100:1:1:1 N H Ru N H R' H C O R
SLIDE 2
SLIDE 3
LIGAND ACCELERATION IN HYDROGENOLYSIS OF EPOXIDES
2-propanol H2 + Cp*RuCl(cod) ligand KOH 10 atm 30 °C, 2 h ketone:Ru:ligand:KOH = 100:1:1.5:1 OH OH branch/linear NH2 (CH3)2N (C6H5)2P N(CH3)2 NHCH3 (C6H5)2P >99/1 >99/1 (C6H5)2P P(C6H5)2 P(C6H5)3 C6H5CH2NH2 + NH2 (C6H5)2P 89/11 <1% 0% 63% conv 15% 5% <1% ligand: O +
SLIDE 4
HYDROGENOLYSIS OF TERMINAL EPOXIDES
R OH 2-propanol H2 + substrate:Ru:ligand:KOH = 100:1:1.5:1 50 °C, 18 h Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH R 100% conv + R OH 90% conv 100% conv 99% conv 99% conv 10 atm branch/linear 89/11 >99/1 >99/1 98/2 >99/1 C6H5 O O C6H5 O O O C6H5 O substrate: O intact
SLIDE 5
P Ru H N H O Ar Ar
A POSSIBLE MECHANISM
Ru HN P Ar Ar Ru H H
2
N Ar Ar P P Ru HN Ar Ar H H C6H5 O C6H5 C6H5 OH H2 H Cp*RuCl(cod) KOH (C6H5)2P(CH2)2NH2 + +
SLIDE 6
HYDROGENOLYSIS OF (S)-STYRENE OXIDE
OH 2-propanol H2 + 50 °C, 18 h Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH OH 10 atm 0% ee substrate:Ru:ligand:KOH = 100:1:1.5:1 O + 89 : 11 100% conv 96% ee
SLIDE 7
RACEMIZATION OF CHIRAL ALCOHOLS
OH 2-propanol 50 °C, 30 min Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH 0% ee 99% ee OH S/C = 100 solvent effect 2-propanol > ethanol > methanol
SLIDE 8
HYDROGENOLYSIS OF (S)-STYRENE OXIDE IN VARIOUS SOLVENTS
OH solvent H2 + 50 °C Cp*RuCl(cod) (C6H5)2P(CH2)2NH2 KOH OH 10 atm substrate:Ru:ligand:KOH = 100:1:1.5:1 O + 96% ee solvent conv, % branch/linear ee, % time, h 2-propanol methanol ethanol 1 2 18 79 64 64 89/11 90/10 96/4 81 84
SLIDE 9