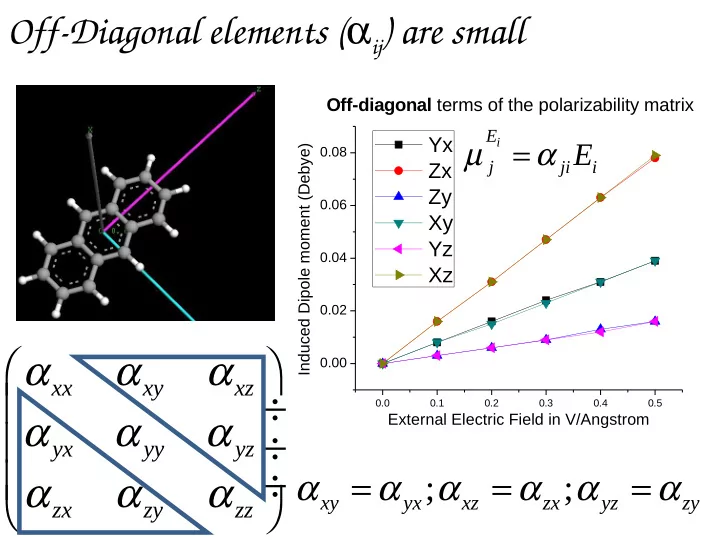
SLIDE 1 0.0 0.1 0.2 0.3 0.4 0.5
0.00 0.02 0.04 0.06 0.08
Induced Dipole moment (Debye) External Electric Field in V/Angstrom
Off-diagonal terms of the polarizability matrix
Yx Zx Zy Xy Yz Xz
xx xy xz yx yy yz zx zy zz
α α α α α α α α α ÷ ÷ ÷
Off-Diagonal elements (αij) are small
; ;
xy yx xz zx yz zy
α α α α α α = = =
i
E j ji i
E µ α = u r

SLIDE 2 E E Ind
E α
µ µ µ
=
= − = u u r u u r u u r u r
x Ix xx xy xz y Iy yx yy yz zx zy zz z Iz
E E E µ α α α µ α α α α α α µ ÷ ÷ ÷ ÷ ÷ = ÷ ÷ ÷ ÷ ÷ ÷ ÷ ÷ u r u r u r u r u r u r
i i
E E i i ii i
E µ µ α
=
− = u r u r
i i
E E j j ji i
E µ µ α
=
− = u r u r
Average of the electronic Polarizability ~ 1/3 xTrace of matrix – sum of diagonal elements. (considering off-diagonal elements relatively small)
and
i i i i
E E E E i i j j ii i ji i
E E µ µ α µ µ α
= =
= + = + u r u r u r u r
Estimate relative magnitudes of polarizability components

SLIDE 3 Test case using Anthracene: Calculate µind by applying Ex, Ey, Ez
Anthracene molecule aligned along z direction
i
E
µ
= =
u r

SLIDE 4
The Hydrogen Bond The Hydrogen Bond

SLIDE 5
Boiling point of hydrides

SLIDE 6 Vibrational (IR) Spectra can measure the strength
O-H S-H
Frequency of oscillation of a bond is related to bond force constant – measure of bond strength

SLIDE 7
What is going on inside liquid water?
Additional stabilization due to interaction between H atoms of one water molecule with the partially electronegative oxygen atom on another adjacent water molecule

SLIDE 8
Structure of liquid Water and Ice
Commonality – H atom between two oxygen atoms; Distance between the H and two oxygen atoms not the same

SLIDE 9 Not all molecules can form hydrogen bond Molecule should have at least one hydrogen, bonded to a more electronegative atom. H-bond can form between two different molecules
Hydrogen bond involves….
X-H…….Y-Z X-H…….Y-Z
H-bond H-bond donor H-bond acceptor

SLIDE 10 Phenol-water Phenol-methanol
Intra-molecular H-bonding
Inter- and Intra-molecular H-Bonds

SLIDE 11
Hydrogen Bond Distances/Energies: Extremely large range!
O H O O H N N H O N H N O H Cl O H P D H A D H H A A D 1.0 1.0 1.1 1.1 1.0 1.0 1.8 1.9 1.8 1.9 2.2 2.4 2.8 2.9 2.9 3.0 3.2 3.4

SLIDE 12 Under certain conditions an atom of hydrogen is attracted by rather strong forces to two atoms instead of only one, so that it may be considered to be acting as a bond between them.
Linus Pauling (1939)
The hydrogen bond is an attractive interaction between a hydrogen atom from a molecule or a molecular fragment X–H in which X is more electronegative than H, and an atom or a group of atoms in the same or a different molecule, in which there is evidence of bond formation
IUPAC definition (2011)
Concept and definition of H-bonding

SLIDE 13 2 3 4 6 12
( ) A B C D F G V r r r r r r r = − − − − − +
Potential energy function for Hydrogen Bonding A - Electrostatics B - Ion-Dipole C - Dipole-Dipole D - Ion-Induced dipole F - Dipole-Induced dipole & Induced dipole -Induced dipole G - Repulsion Each hydrogen bond is unique Each hydrogen bond is unique
Potential Energy for Hydrogen Bond complicated: poorly understood!

SLIDE 14
Importance of Intermolecular Forces and H-Bonds in Biology

SLIDE 15
3-DStructure of Proteins (Enzymes) held together: Intermolecular forces

SLIDE 16
Secondary structure of Proteins: H-Bonds play major role!

SLIDE 17
Held together by H-bonds!

SLIDE 18
The peptide alpha-Helix

SLIDE 19
The peptide beta-sheets

SLIDE 20
Nucleic acids – Double helical DNA

SLIDE 21
Structure of DNA

SLIDE 22 Base-pairing in nucleic acids
is hydrogen bonded to a base on the
forming a base pair
and G bonds with C forming complementary strands.

SLIDE 23
DNA Stabilization--H-bonding between DNA base pair stacks

SLIDE 24
Strength of Cellulose…..

SLIDE 25
Self-healing Rubber based solely on H-Bonds