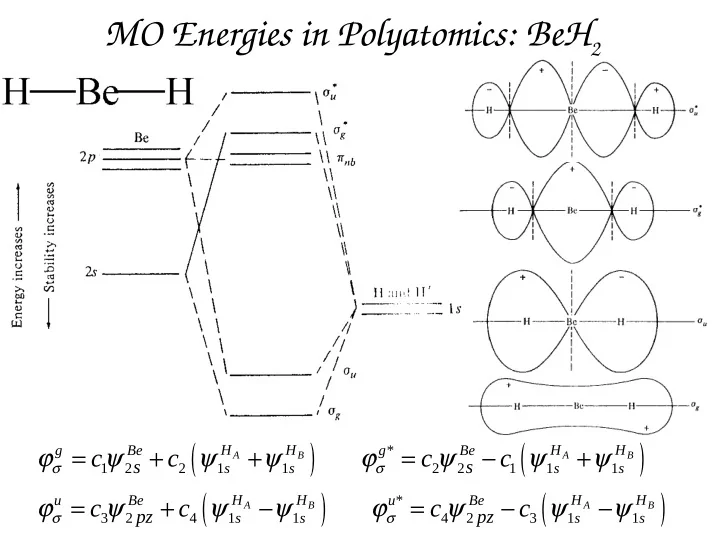
MO Energies in Polyatomics: BeH2
( ) ( ) ( ) ( )
* 1 2 2 1 1 2 2 1 1 1 * 3 2 4 1 1 4 2 3 1 1
A B A B A B A B
H H H H g Be g Be s s s s H H H H u Be u Be s s s s
s s pz pz
c c c c c c c c
σ σ σ σ
Hybrids: Linear Combination of S and P leads to lowering of energy - - PowerPoint PPT Presentation
MO Energies in Polyatomics: BeH 2 ( ) ( ) = + + = + g Be H H g * Be H H c c c c A B A B s s 1 2 2 1 s 1 s 2 2 1 1 s 1 s ( ) ( ) = +
* 1 2 2 1 1 2 2 1 1 1 * 3 2 4 1 1 4 2 3 1 1
A B A B A B A B
H H H H g Be g Be s s s s H H H H u Be u Be s s s s
s s pz pz
σ σ σ σ
Linus Pauling, ~1930
1 1 2 2 1 2 sp h sp h
1 2
sp h sp h
1 1 1 1 2 2 1 3 3
1 2 3
x y x y x y
s p p hy s p p hy s p p hy
30o 30o
30o 30o
30o 30o
30o 30o
30o
30o
2 2 2
1 1 2 3 2 4 5 6 2 7 8 9 sp h sp h sp h x y x y x y
2 2 2
1 1 2 3 2 4 5 6 3 7 8 9
sp h sp h sp h x y x y x y
2 2 2 1 4 7 1 4 7 2 1 5 8 6 9
2 2 2 1 2 3 2 2 2 4 5 6 2 2 2 7 8 9 2 2 2 2 5 8 2 2 2 3 6 9
2 2 2
1 2 3 4 5 6 7 8 9
1 2 3
sp sp sp x y x y x y
h h h
1 4 2 5 3 6 4 7 5 8 6 9 7 1 8 2 9 3
i j
2 2 2
1 2 3
sp sp sp x y x y x y
h h h
s p p s p p s p p
3 3 3 3
1 2 2 4
y
sp h sp h p sp h sp h x y z x z x y z x y z
3 3 3 3
1 1 2 3 4 2 1 5 7 8 2 1 9 10 11 4 1 12 13 14
y
sp h sp h p sp h sp h x y z x z x y z x y z
s p p p s p p s p p p s p p p
1 2 3 4
y
h h p h h x y z x z x y z x y z
s p p p s p p s p p p s p p p