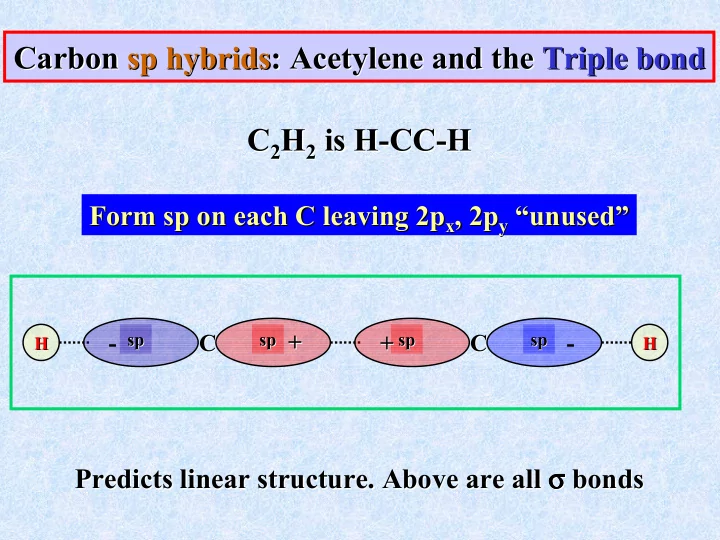
C C C C
H H H H
Carbon Carbon sp sp hybrids hybrids: Acetylene and the : Acetylene and the Triple bond Triple bond C C2
2H
H2
2 is H
is H-
- CC
CC-
- H
H
Form Form sp sp on each C leaving 2p
- n each C leaving 2px
x, 2p
, 2py
y “unused”
“unused” Predicts linear structure. Above are all Predicts linear structure. Above are all σ σ σ σ σ σ σ σ bonds bonds
sp sp sp sp sp sp sp sp
+ +
- +