SLIDE 1
Trace Elements in igneous petrology
- Abundances of trace elements are used to test petrogenetic hypotheses
- No universal definition of TE: Concentration usually less than 100 ppm, often < 10 ppm
- Useful trace elements:
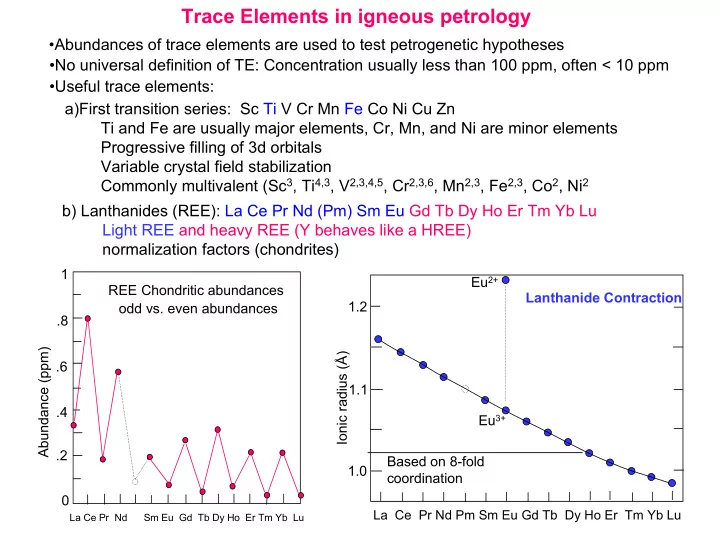
a)First transition series: Sc Ti V Cr Mn Fe Co Ni Cu Zn Ti and Fe are usually major elements, Cr, Mn, and Ni are minor elements Progressive filling of 3d orbitals Variable crystal field stabilization Commonly multivalent (Sc3, Ti4,3, V2,3,4,5, Cr2,3,6, Mn2,3, Fe2,3, Co2, Ni2 b) Lanthanides (REE): La Ce Pr Nd (Pm) Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Light REE and heavy REE (Y behaves like a HREE) normalization factors (chondrites)
Abundance (ppm) 1 .8 .6 .4 .2
La Ce Pr Nd Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
- dd vs. even abundances