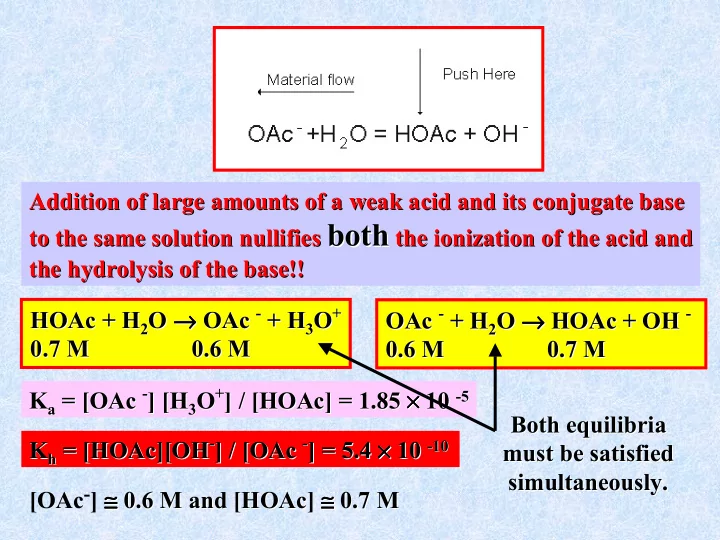
Addition of large amounts of a weak acid and its conjugate base Addition of large amounts of a weak acid and its conjugate base to the same solution nullifies to the same solution nullifies both
both the ionization of the acid and
the ionization of the acid and the hydrolysis of the base!! the hydrolysis of the base!! OAc OAc -
- + H
+ H2
2O
O → → → → → → → → HOAc HOAc + OH + OH -
- 0.6 M 0.7 M
0.6 M 0.7 M K Ka
a = [
= [OAc OAc -
- ] [H
] [H3
3O
O+
+] / [
] / [HOAc HOAc] = 1.85 ] = 1.85 × × × × × × × × 10 10 -
- 5
5
[OAc-] ≅ ≅ ≅ ≅ ≅ ≅ ≅ ≅ 0.6 M and [ 0.6 M and [HOAc HOAc] ] ≅ ≅ ≅ ≅ ≅ ≅ ≅ ≅ 0.7 M 0.7 M HOAc HOAc + H + H2
2O
O → → → → → → → → OAc OAc -
- + H
+ H3
3O
O+
+
0.7 M 0.6 M 0.7 M 0.6 M K Kh
h = [
= [HOAc HOAc][OH ][OH-
- ] / [
] / [OAc OAc -
- ] = 5.4
] = 5.4 × × × × × × × × 10 10 -
- 10
10