SLIDE 1
Page 6 of 21
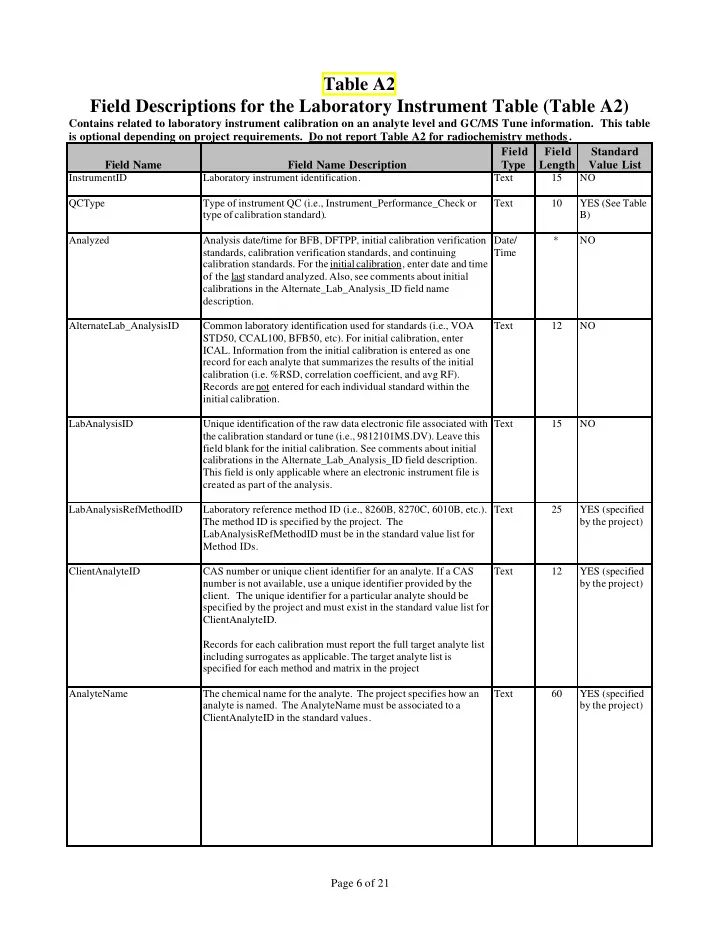
Table A2 Field Descriptions for the Laboratory Instrument Table (Table A2)
Contains related to laboratory instrument calibration on an analyte level and GC/MS Tune information. This table is optional depending on project requirements. Do not report Table A2 for radiochemistry methods . Field Name Field Name Description Field Type Field Length Standard Value List
InstrumentID Laboratory instrument identification. Text 15 NO QCType Type of instrument QC (i.e., Instrument_Performance_Check or type of calibration standard). Text 10 YES (See Table B) Analyzed Analysis date/time for BFB, DFTPP, initial calibration verification standards, calibration verification standards, and continuing calibration standards. For the initial calibration, enter date and time
- f the last standard analyzed. Also, see comments about initial
calibrations in the Alternate_Lab_Analysis_ID field name description. Date/ Time * NO AlternateLab_AnalysisID Common laboratory identification used for standards (i.e., VOA STD50, CCAL100, BFB50, etc). For initial calibration, enter
- ICAL. Information from the initial calibration is entered as one
record for each analyte that summarizes the results of the initial calibration (i.e. %RSD, correlation coefficient, and avg RF). Records are not entered for each individual standard within the initial calibration. Text 12 NO LabAnalysisID Unique identification of the raw data electronic file associated with the calibration standard or tune (i.e., 9812101MS.DV). Leave this field blank for the initial calibration. See comments about initial calibrations in the Alternate_Lab_Analysis_ID field description. This field is only applicable where an electronic instrument file is created as part of the analysis. Text 15 NO LabAnalysisRefMethodID Laboratory reference method ID (i.e., 8260B, 8270C, 6010B, etc.). The method ID is specified by the project. The LabAnalysisRefMethodID must be in the standard value list for Method IDs. Text 25 YES (specified by the project) ClientAnalyteID CAS number or unique client identifier for an analyte. If a CAS number is not available, use a unique identifier provided by the
- client. The unique identifier for a particular analyte should be