Slide 1 / 142
Properties of Matter and Solutions
Slide 2 / 142 Properties of Matter and Solutions
Pyrite, otherwise known as "fools gold" has fooled many a tourist
- ver the years. Physical and
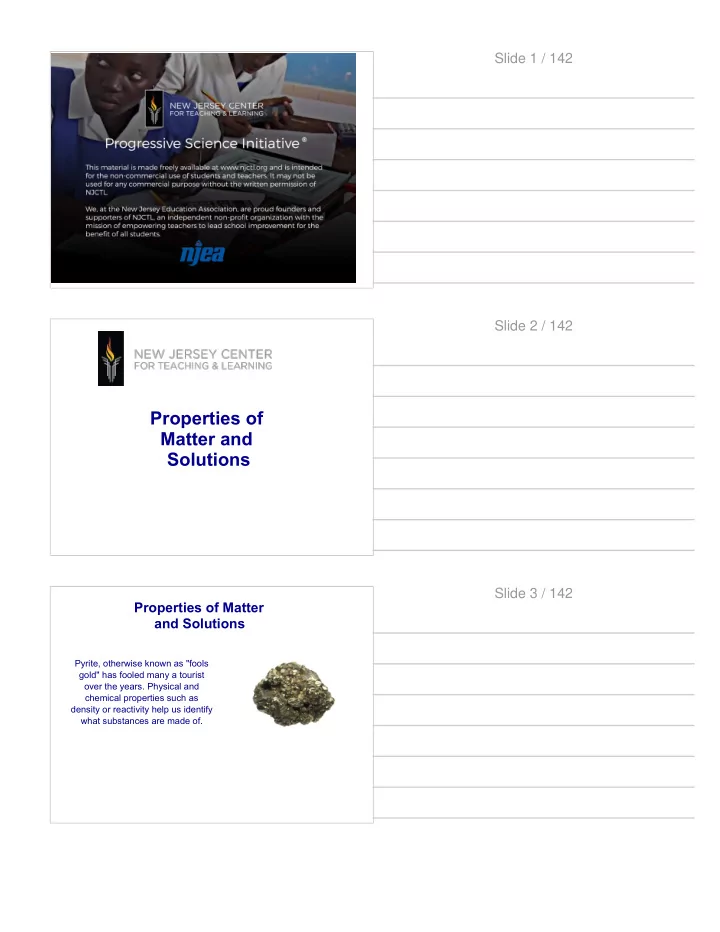
Properties of Matter and Solutions Slide 3 / 142 Properties of - - PDF document
Slide 1 / 142 Slide 2 / 142 Properties of Matter and Solutions Slide 3 / 142 Properties of Matter and Solutions Pyrite, otherwise known as "fools gold" has fooled many a tourist over the years. Physical and chemical properties
We define matter as anything that has mass and takes up space.
Atoms of an element molecules of a diatomic element Molecules of a compound Mixture of elements and a compound
Na Sodium Cu-Copper I-Iodine vapor Al Aluminum foil Mg-Magnesium C-carbon diamond and graphite
CAFFEINE
1 Which of the following would NOT be a
1 Which of the following would NOT be a
[This object is a pull tab]
2 Which of the following is FALSE regarding
2 Which of the following is FALSE regarding
[This object is a pull tab]
electricity
as the Law of Definite Composition.
Some matter can be separated by heat, filtering, or boiling into other substances but did NOT obey the law of definite
NOT pure substances. More on mixtures later! Pure Substance Definitive Composition Examples: gold (Au) pure water (H2O) Mixture Non-definitive composition Examples: steel (Fe, C, Mn, Cr, ...) salt water (H2O, Cl-, Na+, ...)
[This object is a pull tab]
[This object is a pull tab]
It was clear, even to the ancients, that not all matter shares the same characteristics/properties. Substance Property gold lustrous, soft metal, non-reactive, solid at room temperature salt water transparent, liquid at room temperature, could be separated by heat, no definite composition pure water transparent, liquid at room temperature, definite composition, could be separated by electrolysis calcium carbonate solid at room temperature, high melting point, non-lustrous, could be separated by heat
A physical property is a characteristic that can be observed WITHOUT altering the identity of the material. Physical Properties of water water melts at 0 Celsius at standard pressure water is transparent water has a density of roughly 1 g/mL at 25 C water is not soluble (does not dissolve) in gasoline water is colorless Notice all of these properties can be observed without changing the identity of the water - it is still water!
5 Which of the following IS NOT a physical property?
5 Which of the following IS NOT a physical property?
[This object is a pull tab]
6 Which of the following IS a physical property?
6 Which of the following IS a physical property?
[This object is a pull tab]
Chemical changes result in new substances. Includes combustion,
decomposition, etc. Changes in matter that don't change the composition of a substance. Includes changes of state, temperature, volume, etc.
7 Which of the following is NOT a chemical
7 Which of the following is NOT a chemical
[This object is a pull tab]
8 All of the following are physical properties
8 All of the following are physical properties
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
Application When you cook, cheese can be melted or it can be burned. One is a chemical change, the other a physical change. Explain which is which and how you knew! melted burned Melting is a physical change because the cheese has not changed - we know this because we see no evidence of a chemical change (no gas, light, precipitate, color change). However, burning cheese is a chemical change because we clearly see a color change, taste change, production of a gas when you set off the smoke detector!
move for answer
These are properties in which the value is independent of the amount of material. Examples The water is transparent and colorless The melting point of an iron chunk is 1538 Celsius The specific heat (amount of energy required to raise 1 gram by 1 degree celsius) of aluminum is 0.89 J/g*C
Density is an excellent example of an intensive property. No matter the size of the sample, the ratio of the mass to the volume for a given substance is the same. The higher the volume of the sample, the higher the mass will be. mass of water volume of water density of water 19.01 grams 19.03 mL 0.999 g/mL 100.43 grams
Some meteorites found on the earth's surface are made of solid metal like iron. What kind of property - intensive or extensive - do you think would be most useful in identifying the metal in the meteorite? Explain. Intensive properties are unique to each substance so they are better for identifying. You can have 10 grams of just about anything or 5 mL of of just about anything, but only iron has a density of exactly 7.78 g/mL move for answer
11 Which of the following would be an intensive
11 Which of the following would be an intensive
[This object is a pull tab]
12 Tungsten is a substance with an extremely high
12 Tungsten is a substance with an extremely high
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
Credit toTom Greebowe
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
When an ionic substance dissolves in water, the solvent pulls the individual ions from the crystal and solvates them. This process is called dissociation .
_ 2+ _ _ _ _ _ _ 2+ _ _ 2+ _
2+ 2+ 2+ _ _ 2+ _ _ 2+ _ _
A strong electrolyte dissociates completely when dissolved in water. A weak electrolyte only dissociates partially when dissolved in water.
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
_ 2+ _ _
2+
_
solvent water solute
[This object is a pull tab]
∆H1- Separation of solute molecules
∆H2 - Separation of solvent
molecules + ∆H3- Formation of solute-solvent interactions
Separated Separated solvent + solute particles particles
Separated
Solvent + solute particles
Solvent + Solute
Net exothermic process
Reactions, including solution, will occur spontaneously as long as the change in Gibbs Free Energy is negative. When the process, is endothermic (heat is taken in from the surroundings), the increase in enthalpy is offset by an increase in entropy.
Separated Separated solvent + solute particles particles
Separated
Solvent + solute particles Solvent + Solute
Solution
Net endothermic process
Just because a substance disappears when it comes in contact with a solvent, it doesn’t mean the substance dissolved. Dissolution is a physical change — you can get back the original solute by evaporating the solvent. If you can’t, the substance didn’t dissolve, it reacted.
In a saturated solution, the solvent holds as much solute as is possible at that temperature. Dissolved solute is in dynamic equilibrium with solid solute particles.
In supersaturated solutions, the solvent holds more solute than is normally possible at that temperature. These solutions are unstable; crystallization can usually be stimulated by adding a “seed crystal” or scratching the side of the flask.
Click here for a video on Rapid Crystallization
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
Chemists use the axiom “like dissolves like."
Alcohol Solubity in water Solubility in hexane CH
3OH
methanol # 0.12 CH
3CH 2OH
ethanol # # CH
3CH 2CH 2OH
propanol # # CH
3CH 2CH 2CH 2OH
butanol 0.11 # CH
3CH 2CH 2CH 2CH 2OH
pentanol 0.030 # CH
3CH 2CH 2CH 2CH 2CH 2OH
hexanol 0.0058 # solubility expressed in mol/100g solvent # = completely miscible
Polar substances tend to dissolve in polar solvents. Nonpolar substances tend to dissolve in nonpolar solvents.
Hydrogen bonding sites Glucose- has hydroxyl groups and is highly soluble in water Cyclobutane-has no polar OH groups and is essentially insoluble in water
The more similar the intermolecular attractions, the more likely
hydrogen bonding) is very soluble in water, while cyclobutane (which only has dispersion forces) is not.
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
A solubility chart can be used to determine the amount of solute that can be dissolved by a particular solvent at a range of temperatures. The line of a solubility chart represents a saturated
line represents a supersaturated solution at that temperature.
A point above the line represents a supersaturated solution at a specific temperature. The line of a solubility chart represents a saturated solution.
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
The opposite is true of gases. Carbonated soft drinks are more “bubbly” if stored in the refrigerator. Warm lakes have less O2 dissolved in them than cool lakes.
The solubility of liquids and solids does not change appreciably with pressure. The solubility of a gas in a liquid is directly proportional to its pressure.
In general, the solubility of gases in water increases with increasing molar mass. Larger molecules have stronger dispersion forces.
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
Recall that solutions are homogeneous mixtures of two or more pure
throughout the solvent.
State of Solution State of Solvent State of Solute Example Gas Gas Gas Air Liquid Liquid Gas Oxygen in water Liquid Liquid Liquid Alcohol in water Liquid Liquid Liquid Salt in water Solid Solid Gas H2 in Palladium Solid Solid Liquid Hg in Silver Solid Solid Solid Silver in Gold
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
2H3O2 is combined with
2H3O2 is combined with
[This object is a pull tab]
[This object is a pull tab]
Since both moles and mass do not change with temperature, molality (unlike molarity) is not temperature-dependent.
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
Because of solute-solvent intermolecular attraction, higher concentrations of nonvolatile solutes make it harder for solvent to escape to the vapor phase. Therefore, the vapor pressure
that of the pure solvent.
Solvent alone Solvent + Solute
Pressure Temperature 1 atm
We said earlier that colligative properties depend only on the number of solute particles present , not on the identity of the solute particles. However, it's important to note that it's the number of particles in solution, not the number of particles before they are dissolved. If a solute ionizes, you can get more particles in solution than you started with...depending on the substance.
For instance, 1 mol NaCl becomes 2 moles of particles in solution: 1 mol Na+ + 1 mol Cl- 1 mol CaCl2 becomes 3 moles in solution: 1 mol Ca+ + 2 mol Cl- 1 mol C6H12O6 (glucose) stays 1 mol since it doesn't disassociate, it's stays a single molecule because it is a molecular compound. So in terms of colligative properties; you get about three times the effect with CaCl2 (and two times the effect with NaCl) than you do with C6H6.
2O
2O
[This object is a pull tab]
3)2
3
3)2
3 [This object is a pull tab]
3)2
3)2
[This object is a pull tab]
A 0.25 M glucose, C
6H12O6
B 0.50 M glucose
C 0.50 sucrose, C12H22O11 D
1.0 M sucrose
E All of these aqueous solutions have equal vapor pressure.
A 0.25 M glucose, C
6H12O6
B 0.50 M glucose
C 0.50 sucrose, C12H22O11 D
1.0 M sucrose
E All of these aqueous solutions have equal vapor pressure.
[This object is a pull tab]
A 0.75 M glucose, C
6H12O6
B 0.50 M glucose
C 0.25 M sucrose, C12H22O11 D
0.50 M sucrose
E All of these aqueous solutions have equal vapor pressure.
A 0.75 M glucose, C
6H12O6
B 0.50 M glucose
C 0.25 M sucrose, C12H22O11 D
0.50 M sucrose
E All of these aqueous solutions have equal vapor pressure.
[This object is a pull tab]
A pure water
B 1.0 m sucrose (aq) C 1.0-m NaCl (aq) D 1.0-m HCl (aq) E 1.0-m CaCl2 (aq)
A pure water
B 1.0 m sucrose (aq) C 1.0-m NaCl (aq) D 1.0-m HCl (aq) E 1.0-m CaCl2 (aq)
[This object is a pull tab]
A pure water
B 1.0 m sucrose (aq) C 1.0-m CaCl2 (aq) D 1.0-m HCl (aq) E 1.0-m KCl (aq)
A pure water
B 1.0 m sucrose (aq) C 1.0-m CaCl2 (aq) D 1.0-m HCl (aq) E 1.0-m KCl (aq)
[This object is a pull tab]
A pure water
B 1.0 m sucrose (aq) C 1.0-m NaCl (aq) D 1.0-m HCl (aq) E 1.0-m CaCl2 (aq)
A pure water
B 1.0 m sucrose (aq) C 1.0-m NaCl (aq) D 1.0-m HCl (aq) E 1.0-m CaCl2 (aq)
[This object is a pull tab]
A pure water B 1.0 m sucrose (aq) C 1.0-m NaCl (aq) D
1.0-m HCl (aq) E 1.0-m CaCl2 (aq)
A pure water B 1.0 m sucrose (aq) C 1.0-m NaCl (aq) D
1.0-m HCl (aq) E 1.0-m CaCl2 (aq)
[This object is a pull tab]
A pure water
B 0.20-m glucose (aq) C 0.20-m KBr (aq) D 0.20-m HCl (aq) E 0.20-m AlCl3 (aq)
A pure water
B 0.20-m glucose (aq) C 0.20-m KBr (aq) D 0.20-m HCl (aq) E 0.20-m AlCl3 (aq)
[This object is a pull tab]
A pure water
B 0.15-m Mg(NO3)2 (aq) C 0.15-m glucose(aq) D 0.15-m NaF (aq) E 0.15-m HBr (aq)
A pure water
B 0.15-m Mg(NO3)2 (aq) C 0.15-m glucose(aq) D 0.15-m NaF (aq) E 0.15-m HBr (aq)
[This object is a pull tab]
[This object is a pull tab]
A increases, increases B increases, decreases C decreases, increases D unaffected, decreases E decreases, decreases
A increases, increases B increases, decreases C decreases, increases D unaffected, decreases E decreases, decreases
[This object is a pull tab]
[This object is a pull tab]
[This object is a pull tab]
Credit to Tom Greenbowe
Some substances form semipermeable membranes, allowing some smaller particles to pass through, but blocking other larger particles. In biological systems, most semipermeable membranes allow water to pass through, but solutes are not free to do so.
If the solute concentration outside the cell is greater than that inside the cell, the solution is hypertonic . Water will flow out of the cell, and crenation results.
If the solute concentration
inside the cell, the solution is hypotonic. Water will flow into the cell, and hemolysis results.
[This object is a pull tab]
[This object is a pull tab]