Physical properties of magmas (density (ρ), viscosity (η), thermal conductivity (κ): (control flow rates, cooling rates, eruption rates etc.)
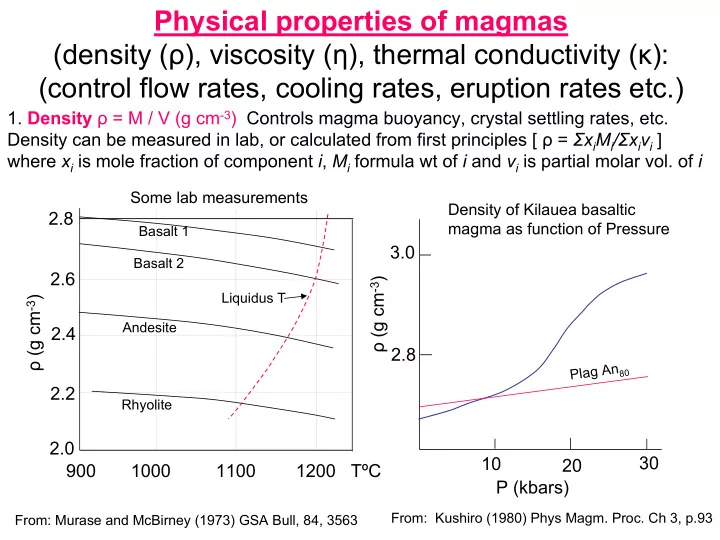
- 1. Density ρ = M / V (g cm-3) Controls magma buoyancy, crystal settling rates, etc.
Density can be measured in lab, or calculated from first principles [ ρ = ΣxiMi/Σxivi ] where xi is mole fraction of component i, Mi formula wt of i and vi is partial molar vol. of i 900 1000 1100 1200 TºC ρ (g cm-3) 2.0 2.2 2.4 2.6 2.8
Basalt 1 Basalt 2 Andesite Rhyolite Liquidus T
Some lab measurements
From: Murase and McBirney (1973) GSA Bull, 84, 3563
ρ (g cm-3) 2.8 3.0 10 20 30 P (kbars)
Density of Kilauea basaltic magma as function of Pressure
Plag An80 From: Kushiro (1980) Phys Magm. Proc. Ch 3, p.93