SLIDE 1
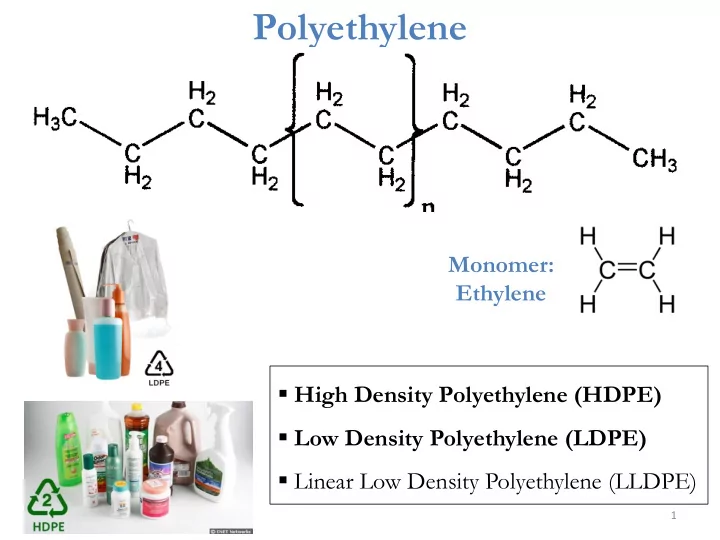
Polyethylene
Monomer: Ethylene
- High Density Polyethylene (HDPE)
- Low Density Polyethylene (LDPE)
- Linear Low Density Polyethylene (LLDPE)
1