Phase Transformations / Hardenability (Jominy EndQuench) Presentation Fall '15 (2151) MECE306 Materials Science Apps Lab 1
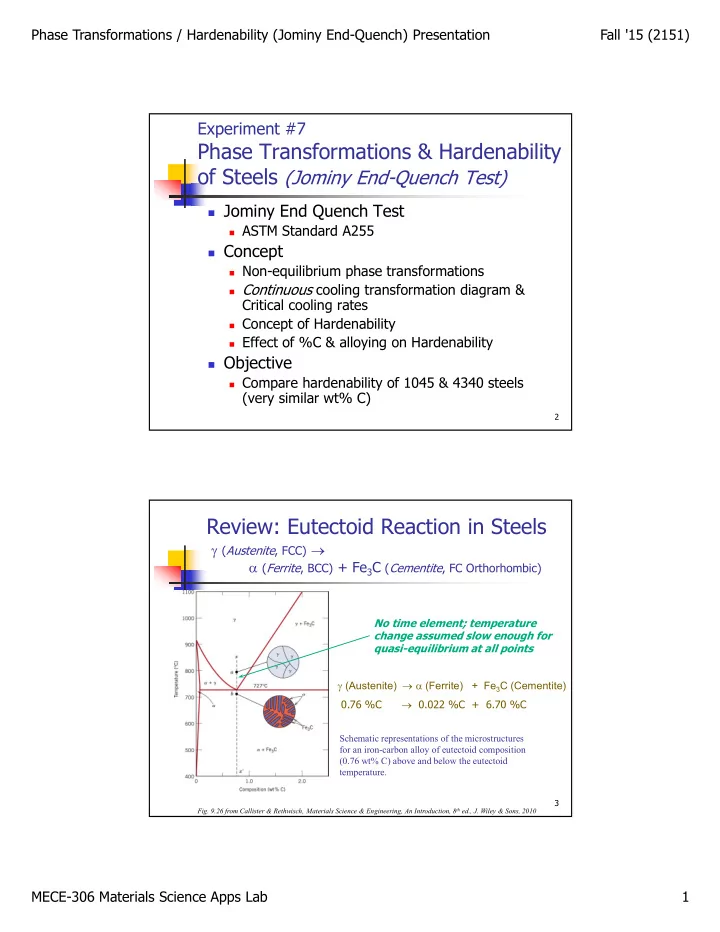
Experiment #7
Phase Transformations & Hardenability
- f Steels (Jominy EndQuench Test)
Jominy End Quench Test
ASTM Standard A255
Concept
Nonequilibrium phase transformations Continuous cooling transformation diagram &
Critical cooling rates
Concept of Hardenability Effect of %C & alloying on Hardenability
Objective
Compare hardenability of 1045 & 4340 steels
(very similar wt% C)
2
Review: Eutectoid Reaction in Steels
γ (Austenite, FCC) → α (Ferrite, BCC) + Fe3C (Cementite, FC Orthorhombic)
γ → α 0.76 %C → 0.022 %C + 6.70 %C
- ! "#$%&%
3