McGuffee & Elcock (2010) Diffusion, crowding & protein stability in a
McGuffee & Elcock (2010) Diffusion, crowding & protein - - PowerPoint PPT Presentation
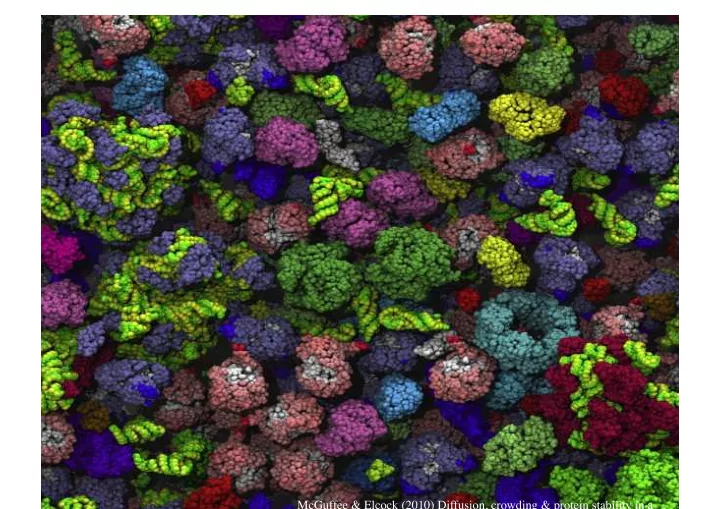
McGuffee & Elcock (2010) Diffusion, crowding & protein - - PowerPoint PPT Presentation
McGuffee & Elcock (2010) Diffusion, crowding & protein stability in a Macromolecular Crowding Molecules and Complexes: E. coli Census Nproteins ~ 3x10^6 Nribosomes ~ 20,000 Nlipids ~ 2x10^7 Spacing for a molecule of given
Macromolecular Crowding
Molecules and Complexes: E. coli Census
- Nproteins ~ 3x10^6
- Nribosomes ~ 20,000
- Nlipids ~ 2x10^7
Spacing for a molecule of given concentration c:
d = c−1 3
Protein Spacing in E. coli
- Estimate
- Average protein-protein distance ~ 10 nm
- 1 mM protein in vitro distance ~ 100 nm
Radius of protein ~ 2nm 1 um
Linked Polymer Network Architecture
Gram positive bacterial wall (atomic resolution) Collagen fibrils Basement membrane Actin web in a miving cell Cellulose network
- f a plant cell
Ligament collagen bundle Axon bundle Intenstinal microvilli
- Effect on binding and interaction
- Difference between cells and dilute
solutions
Ten Commandments of Enzymology, Amended
Kornberg (2003) Tr. Biochem. Sci.
- 1. Thou shalt rely on enzymology to resolve
and reconstitute biologic events
- 2. Trust the universality of biochemistry and
the power of microbiology (Escherichia sapiens)
- 3. Not believe something just because you can
explain it
- 4. Not waste clean thinking on dirty enzymes
- 5. Not waste clean enzymes on dirty substrates
- 6. Use genetics and genomics
- 7. Be aware that cells are molecularly crowded
- 8. Depend on viruses to open windows
- 9. Remain mindful of the power of radioactive
tracers
- 10. Employ enzymes as unique reagents
Arthur Kornberg Nobel Prize for Chemistry (1959) Discovery of DNA polymerase (now known as DNA polymerase I)
Be aware that cells are molecularly crowded
Fish skin keratocyte
Fish Keratocyte Leading Edge
Membrane stripped platinum coated E.M. Front edge: Ordered, Branched Middle zone: Randomly
- verlaid
filaments
Cellular Effects of Crowding
- Equilibrium binding
- Diffusive processes
7.5% 5% 2.5% 0% PEG concentration
ATPase Rate
T4 DNA polymerase clamp-loader proteins
Experimental Measures
- f Diffusivity
BODIPY-FI sizes
Verkman (2002) TiBS
Method 1 Method 2 FCS Time resolved Fluorescence Anisotropy Method 3
Cellular Diffusion Coefficients
Verkman (2002) TiBS
Single Molecule Imaging Membrane Proteins
GFP-Lck Lck Tyrosine kinase Jurkat T-cells TIRF microscopy Anti-T cell receptor Abs stimulate clustering
Douglass & Vale (2005)
Trapped Long distance
Membrane Microdomains
T-cell Signalling domains in CD2-enriched signalling domains Signalling activated by antibody-patch on coverslip
CD2 Lck Merge
Lipid Rafts
GPI-anchored proteins Raft-associated lipids
Mayor Lab, NCBS Bangalore
Effective Diffusion
<d^2> = < [d(t)-d(t+δt)]^2 >, For different values of δt. Plotting d^2 vs δt gives us a profile that can be fitted by <d^2>=2*D*t^α α = 1 for normal diffusion
Bacher, Reichenzeller, Athale et al. (2004) Klopfstein et al. (2002) Cell
Raft targeted Lck-10 GFP Lck GFP
TIRF: At the Surface
Evanescent wave illumination with limited range
Nikon Instruments
Lattice Model of Crowding
Ωv = reaction volume L=ligand no. C=Crowding
- molec. no.
Ligand-Receptor Binding pbound = 1 1+ Ω− L − C L
( )eβΔε L
L << Ω When C increases, pbound increases
ΔεL = εL
b −εL sol
Crowding Changes L-R binding Probability
7.5% 5% 2.5% 0% PEG concentration Binding constant PEG dependent)
Dissociation Rate
Kd = 1 v eβΔe
pbound = L
[ ] Kd
( )
n
1+ L
[ ] Kd
( )
n
v=volume of single lattice site Δε = binding energy
PEG as Crowdant
T4 atpase data, PEG size (12 kDa) << Protein size (164 kDa) Ω large boxes r small boxes in each large box
Crowdant Smaller than Ligand
Binding
Where, volume fraction of the crowding molecules in solution Assuming L << Ω And (N+r)!/N! ~ Nr
φC = C rΩ
pbound = 1 1+ Ω L (1− φC )reBΔε L
Dissociation Constant
Volume fraction dependent dissociation constant Kd
Kd φC
( )
Kd φC = 0
( )
= 1− φC
( )
r
Kd = 1 v eβΔe
Factors Affecting Crowding
Osmotic Pressure and Crowding
Osmotic pressure due to excess Hemoglobin p = pressure v=volume of single box in lattice [H]=concentration of Hb molecules=H/Ωv
p = − kBT v ln(1−[H]v)
Crowding and Osmotic Pressure
Free parameter is v V=5.8 nm Hard sphere gas model Experiment Lattice gas
Hard Sphere Gas Model
Boltzmann 1899 Where x = 4V[H] V=volume of hard sphere
p = kBT[H](1+ x + 0.625x 2 + 0.287x 3 + 0.11x 4)
Next
- Crowded polymers and ordering
- Cytoskeleton
- Motors
- Cells in tissues
- FRAP data
- Paper presentations
2011-03-23
Macromolecular Crowding
- 10-100% of fluid volume of cytoplasm lies
within 1 molecular diameter of the surface
- f fibrous and membraneous structures
- Pores
- Reactant X and pore size comparable
Sieving Effect
Verkman (2002) TiBS
Volume Exclusion
Minton (2001) J. Biol. Chem.
Excluded volume Available volume
Minton (2001) J. Biol. Chem.
Relative Sizes
Volume Available
Effective and actual concentrations vtot=total volume va,i=volume available to species i
γ i ≡ ai ci
( ) = vtot va,i
( )
Haemoglobin Concentration
Normal RBC Concentration ~ 300gm/L
2011-03-29
Forces due to Volume Exclusion
Large particle near surface Two large particles in solution Two rod-like molecules in solution Depletion forces Volume available to smaller molecules increases
Origin of Depletion Forces
R= radius of large disk r = radius of small disk Surface 2D geometry Find area available to small disks, as a function of distance z between large disk and surface
Excluded Volume Interactions
R= radius of large disk r = radius of small disk Surface 2D geometry z=distance between large disk and surface
Area available to small disks Entropy increases
Free Energy Change
No conventional forces- van der Walls, electrostatics, etc. Free energy change by change in entropy is: Vbox = volume of box, Vex=excluded volume, v=volume
- f unit cell, N=no. of SMALL MOLECULE particles
Gex = −NkBT ln Vbox −Vex v + NkBT ln Vbox v
Free Energy
If Vex << Vbox, approximate ln(1+x) ≈ ln(x) If 2 large particles overlap excluded volumes, Vex increases entropy of small particle ~ ideal gas (osmotic) pressure of small particles in box
Gex = NkBT Vex Vbox
NkBT Vbpx
Depeletion Force
Volume and Force
Total excluded volume Volume of spherical cone Volume cone Overlap Depletion Force
Vex = 2⋅ 4π 3 R + r
( )
3 −Voverlap
Vsphericalcone = 2π 3 R + r
( )
2 ⋅ R + r − D 2
( )
Vcone = π 3 D 2 R + r
( )
2 − D 2
( )
2
[ ]
Voverlap = 2π 3 R + r + D 2
( )
2 2R + 2r + D 2
( )
Fdepletion = −∂Gex ∂D = −pπ R + r
( )
2 − D2
4
p = nkBT , n=N/Vbox, and distance 2R<D<2(R+r)
Depletion Force Measurement
2 beads, R=625 nm, Move in line Depletion agent- Phage λ DNA r=500 nm
DNA conc. DNA conc. DNA conc.
p(D) ∝e−βGex (D)
Gex(D) = pVoverlap
Entropic Ordering
Rods: Filamentous viruses Spheres
Volume Exclusion
Mutual exclusion v=volume occupied N=number of macromolecules Ω=total number of boxes N=no. of macromolecules In absence of excluded volume Zex(N) = Ω! N! Ω− N
( )!
Znex(N) = ΩN N!
Free Energy for Excluded Volume
Free energy Using stirling’s approximation, and assuming Ω >> N, and
G = −kBT lnZ
ΔGex = Gex − Gnex = −kBT ln Zex Znex
1− N Ω
( )
Ω ≈ e−N
Zex Znex ≈ 1− N Ω
N
ΔGex = −NkBT ln 1− N Ω ≈ kBT N 2 Ω
Polymers, Crowding and RW Model
- Random walk model ignores self-avoidance
- Size of macro-molecule like DNA ~
N=number of segments, a=persistence length
Na2
Competing effects:
- Entropy makes chain compact
- Self-avoidance swells the chain
Random Walk Polymer
Free energy R=radius of polymer SRW(R)=random walk entropy of chain of length R Entropy from probability distribution P(R;N)
G(R) = −TSRW (R) + Gex(R)
SRW (R) = kBT lnP(R;N) + const = −kB 3R2 2Na2 + const
Excluded volume
- Assume polymer to be gas with hard cylinders
- f length a and diameter d
- Mutual orientation angle θ decides excluded
volume v = 2da2 sinθ
- For d<<a
- Averaging sin(θ) over all orientations gives
estimate for excluded volume
- Free energy
- Volume fraction of N hard cylinders
πa2d 2
Gex = kBTNφ
φ(R) = N 1 2 πa2d 1 2 πR3 = N 3a2d 8R3
Free Energy Difference
- Since
- Flory’s estimate of free energy of polymer
- Size of chain
ΔGex ≈ kBT N 2 Ω = kBTNφ Gex(R) = kBTN 2 3a2d 8R3 G flory(R) = kBT 3R2 2Na2 + kBTN 2 3a2d 8R3
Rflory = 3 8 a4d
1/ 5
N 3/ 5
RW vs. SAW
- Power scaling RW (N1/2), SAW (N3/5)
- Short polymers RW still valid
For DNA d~2nm, a=100nm For N<<16(d/a)=40,000, G self avoid < RW entropy L=Na=40,000x100 nm (kuhn length) ~ 16 µm
Gex = kBT 3 8 d a N
1 2
SRW = 3 2 kBT
Protein Folding & Crowding
Chaperones
Diffusion in Crowded Environs
Diffusion in Crowded Environments
- For single time step t=τ
- After time t, steps N=t/τ, so
- Diffusion coefficient
For a random walker
x 2 τ = a2 ⋅ pright + a2 ⋅ pleft + 0⋅ pstay = a2(1− φ) x 2 t = t τ x 2 τ = a2 τ (1− φ)t
D = D0(1− φ)
D0 = a2 2τ
FITC-Aldolase diffusing
Aldolase BSA Ovalbumin
Self-Diffusion and Crowding
NEXT
- Cytoskeleton and motors
- Cells
- Development