SLIDE 1
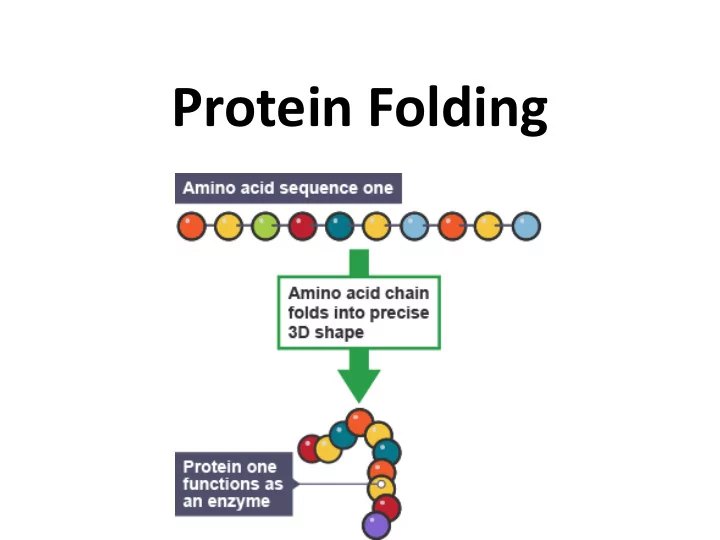
Protein Folding
- Proteins have unique 3-dimensional shapes
created by the twisting or folding of one or more polypeptide chains
- The structure of a protein enables it to
Protein Folding Protein Folding Proteins have unique 3-dimensional - - PowerPoint PPT Presentation
Protein Folding Protein Folding Proteins have unique 3-dimensional shapes created by the twisting or folding of one or more polypeptide chains The structure of a protein enables it to recognize and bind to other molecules Protein