Hydrogen Energy Levels
- 20
- 10
10 20
- 14
- 12
- 10
- 8
- 6
- 4
- 2
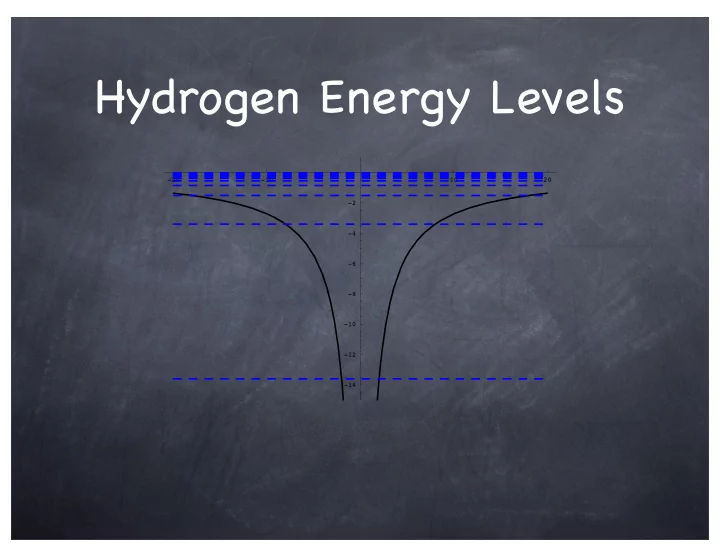
Hydrogen Energy Levels -20 -10 10 20 -2 -4 -6 -8 -10 -12 - - PowerPoint PPT Presentation
Hydrogen Energy Levels -20 -10 10 20 -2 -4 -6 -8 -10 -12 -14 hydrogen wavefunctions 5 10 15 20 -2 -4 -6 -8 -10 -12 -14 n=3 1 0.5 5 10 15 20 25 30 -0.5 -1 -1.5 -2 -2.5 -3 100 200 210 211 300 310 311 320
10 20
5 10 15 20
5 10 15 20 25 30
0.5 1
r × p Lx = ypz − zpy, Ly = zpx − xpz, Lz = xpy − ypx px →
i ∂ ∂x
py →
i ∂ ∂y
pz →
i ∂ ∂z
[Lx, Ly] = [ypz − zpy, zpx − xpz] = [ypz, zpx] − [ypz, xpz] − [zpy, zpx] + [zpy, xpz] = ypx [pz, z] + pyx [z, pz] = i (xpy − ypx) = i Lz [Ly, Lz] = i Lx, [Lz, Lx] = i Ly
L = L2 = L2
x + L2 y + L2 z
x, Lx
y, Lx
z, Lx
Ly [Ly, Lx] + [Ly, Lx] Ly +Lz [Lz, Lx] + [Lz, Lx] Lz = Ly (−iLz) + (−iLz) Ly +Lz (iLy) + (iLy) Lz =
L
[AB, C] = ABC − CAB = ABC − ACB + ACB − CAB = A[B, C] + [A, C]B