SLIDE 1
- 2. This is known as the simple “Basalt” system since this quaternary system represents a simplified
representation of basalt compositions. All the key minerals are present (including En) and, while it is obviously a model system, it illustrates most of the key phase equilibria in basalts. It has been studied extensively and is now well known at pressure ranging from 1 atm to 30 kilobars as the result of the work of a number of experimentalists, notably John Longhi and Dean Presnall.
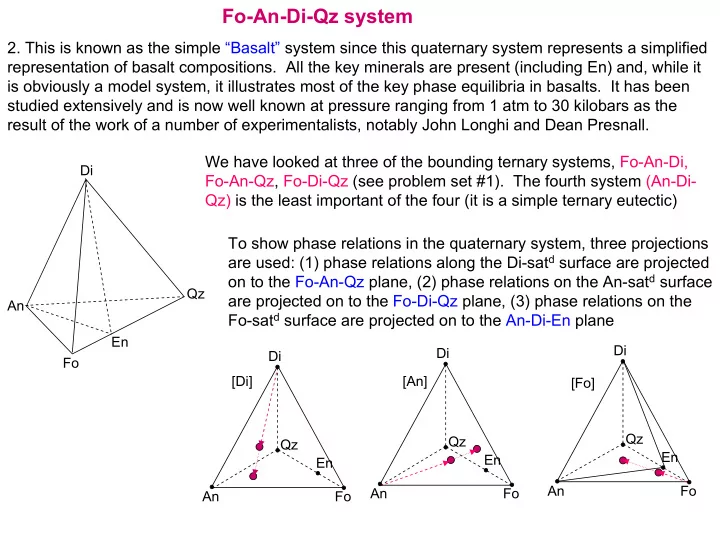
An Fo Qz Di En An Fo Qz Di En [Fo] An Fo Qz Di En [Di] [An] Di An Fo Qz En
We have looked at three of the bounding ternary systems, Fo-An-Di, Fo-An-Qz, Fo-Di-Qz (see problem set #1). The fourth system (An-Di- Qz) is the least important of the four (it is a simple ternary eutectic) To show phase relations in the quaternary system, three projections are used: (1) phase relations along the Di-satd surface are projected
- n to the Fo-An-Qz plane, (2) phase relations on the An-satd surface