Ex Extract ractio ion The British Pharmacopoeia contain a number - - PowerPoint PPT Presentation
Ex Extract ractio ion The British Pharmacopoeia contain a number - - PowerPoint PPT Presentation
Ex Extract ractio ion The British Pharmacopoeia contain a number of extraction of vegetable drugs. Also, the starting material for some products such as steroids may come from natural sources Extraction can be defined as: the removal of
The British Pharmacopoeia contain a number of extraction
- f vegetable drugs. Also, the starting material for some
products such as steroids may come from natural sources Extraction can be defined as: the removal of soluble material from an insoluble residue, either liquid or solid, by treatment with a liquid solvent. It is, therefore, a solution process. The controlling factor in the rate of extraction is normally the rate of diffusion of the solute through the liquid boundary layer or layers at the interface.
Ex Extract ractio ion
LIQ IQUID ID/LIQ IQUID ID EX EXTRACTIO ION Principle of liquid/liquid extraction
1.A solution of the substance to be extracted is brought
into contact with another solvent for the substance that is immiscible with the first solvent.
- 2. A concentration gradient is set up between the phases,
and mass transfer will occur until an equilibrium is established.
3.The distribution of the solute depending on the
distribution coefficient.
- 4. As the process is controlled by mass transfer, the
liquids must be thoroughly mixed to give a large enough surface area for contact, sufficient time for equilibrium to be set up and, finally, the liquids are separated.
Example of small-scale use of liquid/liquid extraction is the purification procedures used in alkaloidal assays.
- This can be achieved using separating funnel in the
laboratory (Small Scale) and using stirred tanks in large- scale .
- Columns may be used for extraction process.
- In pharmaceutical processes centrifugal extractors,
which ensure contact of the liquids in countercurrent flow, followed by rapid separation, are commonly employed.
The liquids enter through the shaft, where the light liquid
being taken to the periphery of the drum, while the heavy liquid enters at the axis of the shaft.
Centrifugal action due to the rotation of the drum causes
the heavy liquid to move towards the outside, so that the light liquid is displaced towards the shaft, both escaping again through the shaft.
The The Po Podbielniak elniak Ex Extr trac actor tor Principle of Operation The body of which consists of a
drum mounted on a horizontal shaft and containing a number
- f
concentric perforated cylinders.
The ability to deal with liquids that tend to emulsify
- r which have only a small
difference in density.
Liquid hold-up is low and
processing time is short.
Advantage of the use of centrifugal force
Mass transfer coefficients are high and the perforated
cylinders causing good dispersion and contact as the liquids move in countercurrent flow.
Disadvantages
The equipment is expensive, but is justified by the high cost
- f many of the medicaments
Application
The method is used widely in the purification of many antibiotics where multistage extraction and centrifugal extractors are used for minimum of decomposition.
Leaching is the extraction of a soluble constituent from a
solid by a solvent. Although fewer preparations are now made by extracting the active constituents of vegetable drugs, the process is still of considerable importance and has more problems than liquid/liquid extraction.
SO SOLID ID/LIQ IQUID ID EX EXTRACT CTIO ION
Difficult culties ies of
- f Extract
racting ng Vege getable ble Drugs
- There are great variations in the characters of vegetable
drugs, some are soft and spongy and can be extracted easily in the whole state, while others are very hard and tough (certain seeds).
- There
are considerable differences in the active constituents to be extracted; alkaloids, glycosides, tannins, resins or oils.
- Different forms of insoluble matter may affect the
extraction process, as the cellulose of the cell structure, and proteins or carbohydrates such as starch.
- In many drugs the active constituent is not the only
soluble material present and a drug may contain a small proportion of an alkaloid with a large proportion of sugars. In this case, a solvent is chosen that is selective to dissolve the alkaloid only.
- Wet vegetable material is an excellent medium for
microbial growth and, if allowed to occur, this may lead to loss of constituents and deterioration of the product. Thus the solvent must have suitable preservative properties.
All extraction processes have common stages
Suitable size reduction of the drug. Penetration of the drug by the solvent. Solution of the soluble matter within the cells. The escape of the dissolved material through the cell walls
and through the solvent boundary layer surrounding the particles of the drug.
Finally, separation of the solution and the exhausted drug.
SIZE REDUCTION OF THE DRUG
The appropriate degree of size reduction will cause large enough surface area for adequate mass transfer: Cause some of the cells of the drug to be broken to assist penetration of the solvent and escape of soluble matter. Provide a particle size that will not result in a very long path for the solvent and the soluble matter. This distance will be equivalent to the radius of a sphere,
- r the shortest distance from any point in the interior to
the surface in particles of irregular shape.
For excellent mass transfer, the maximum surface area should be obtained till the individual cells, but in practice this is not possible because: It would be very difficult to size reduce materials to this extent. A prolonged size reduction process may lead to decomposition
- f
constituents
- r
loss
- f
volatile materials. A suspension of extremely fine particles would form an unfilterable slime which is very difficult to be separated in the final stages.
Breakage of all cells releases the entire cell contents, including inert materials, so that dry extracts are diluted. Therefore it is preferable to keep many of the cell walls unbroken to act as a filter for the retention of insoluble matter or colloidal materials.
The degree of size reduction to be used will depend on the botanical structure of the drug:
Ranging from a sliced for soft drugs (such as gentian) Coarse for materials such as cascara or belladonna Moderately fine Powder for a hard and woody drug as
ipecacuanha. It has been shown that, if dry extracts are made from drugs such as belladonna or stramonium, it is possible to obtain a greater total extractive when finer grades of powder are used, but a higher proportion of alkaloid when a moderately Coarse Powder are used.
PENETRATION OF THE SOLVENT INTO THE DRUG
A drug in the dry state is porous due to shrinkage, and the pores contain air that must be displaced as the solvent enters into the pores and penetrates into the cells. When the dry drug is moistened, the tissues swell, the amount of swelling is variable, being greatest with liquids when hydroxyl groups form a great part of the molecule. Thus, water causes considerable swelling, while is much less with ethanol. Glycerol will lead to swelling as that
- btained with water but the greater viscosity of glycerol
causes the liquid to be taken up much more slowly.
The swelling continues until the pressure caused by the liquid layers cause distension and bursting of thin-walled cells that have taken up liquid by osmosis. As the solvent must displace air from pores, this can be aided by using a vacuum pump. When the pressure is reduced the atmosphere forces solvent into the drug, which facilitate penetration.
SOLUTION OF CONSTITUENTS
Once the solvent has penetrated into the cells, solution of the constituents takes place and the rate of solution is increased by elevation of temperature.
ESCAPE OF THE SOLUTION FROM THE CELLS
During the size reduction process, some cells are broken
- pen, while others are damaged or distorted and, where this
- ccurs, the escape of the solution is not hindered.
However many cells remain whole, so that the soluble matter must pass through the walls. Thus, the rate of diffusion will depend on:
- The presence of a suitable concentration gradient through
the boundary layer
- The thickness of the boundary layer
- The diffusion coefficients of the solutes in the solvent.
Factors affecting the rate of extraction and controlling mass transfer:
- 1. Where the drug is immersed in a quantity of solvent:
(a)By agitating the mixture occasionally, which disperses local concentrations of the solution, thus increasing the concentration gradient. (b)By agitating the solvent and the drug continuously, which reduces the thickness of the boundary layer. (c)By suspending the drug in a cloth bag near to the surface
- f the liquid.
As the constituents dissolve, the density of the solution increases, so that convection currents are established, leading to circulation of the solution.
- 2. If solvent flows past the particles:
(a) The flow replaces the solution by pure solvent, causing an increase in the concentration gradient. (b) The spaces between the particles of drug form passages through which the solvent flows; due to the small size of these capillaries, the velocity of the solvent is sufficient to reduce the boundary layers, so increasing the concentration gradient. In both 1 and 2, the extraction can take place at elevated temperatures, if the material is thermostable and if the solvent is not affected by heating.
- 3. When elevated temperatures can be used, there are a
number of advantages leading to increased extraction rates: (a) The viscosity of the solvent is decreased, which reduces the boundary layers. (b) Convection currents have a similar effect to agitation during extraction. (c) The solubility of the constituents is increased as the temperature is raised.
Extraction will never be complete if the drug is simply immersed in a bulk of solvent, since an equilibrium is set up between the solution in the cells and the free solution. On the other hand, extraction is complete in methods where the drug forms a packed bed with the solvent flowing through it, that fresh solvent is continually displacing the solution.
SEPARATION OF SOLUTION AND EXHAUSTED DRUG
Since the drug absorbs solvent, there is a residue of soluble constituents in this solvent, the drug is subjected to pressure to expel as much of the solution as possible.
Pro roperties erties of
- f Sol
- lven
vents ts Used for
- r Ext
xtra racting cting Dru rugs
As many products made by extraction are intended for internal administration, some restrictions are require on the solvents that can be used. The ideal solvent would be:
- 1. Cheap.
- 2. Non-toxic.
- 3. Chemically and physically inert
- 4. Not too volatile and non-inflammable.
- 5. Selective, to remove the desired active constituents, with
the minimum amount of the inert materials.
Water and ethanol their mixtures are most commonly used;
WATER AS A SOLVENT
Water is a solvent of proteins, colouring matters, gums, anthraquinone derivatives, most alkaloidal salts, glycosides, sugars and tannins. In addition, water will dissolve enzymes, many organic acids, most organic salts, and small proportions of volatile
- ils.
Waxes, fats, fixed oils and most alkaloids (that is, the free bases) are insoluble.
Advantages
(a) Cheap. (b) Has a wide solvent action. (c) Non-toxic. (d) Non-inflammable.
Disadvantages
(a) It is not selective; the list given above shows that it dissolves a wide range
- f substances.
(b) Bacteria grow in aqueous media. (c) Water promotes hydrolysis of many substances and allows enzyme action to take place. (d) Concentration of an aqueous extractive involves the use
- f more heat than other solvents, due to the high latent
heat of vaporisation of water.
ETHANOL AS A SOLVENT
Ethanol is a solvent of alkaloids, alkaloidal salts, glycosides, volatile oils and resins, many forms of colouring matter (chlorophyll), tannins, anthraquinone derivatives, and many
- rganic acids and salts.
Ethanol does not dissolve albuminous matter, gums, waxes, fats, and most fixed oils; Sucrose is insoluble, but certain other sugars present in drugs (dextrose, for example) are soluble in ethanol.
Advantages
(a)It is selective; for example, in a drug containing gum, albuminous matter and a glycoside or an alkaloidal salt, ethanol in a suitable dilution with water would dissolve only the glycoside or alkaloidal salt, whereas water dissolve all these constituents. (b) Moulds and bacteria cannot grow in solutions containing 20 per cent or more of ethanol. (c) It is non-toxic in the quantities of medicinal doses.
(d) It is neutral and compatible with other products. (e) The presence of ethanol in preparations advance rapid absorption of the constituents. (f) The heat of vaporisation of ethanol is less than that of water, so that less heat is used in the concentration of ethanol solutions. (g) Ethanol is miscible with water in all proportions;
Disadvantage
Expensive
The final selection of the solvent is based largely on the characters of the constituents, both active and inert, so that a knowledge of the pharmacognosy of the drug is important.
Ext xtra ract ctio ion Me Method
- ds
The extraction processes may be divided into two main groups. Maceration methods Based on the immersion of the drug in a bulk of solvent. Percolation methods Depend on the flow of solvent through the powdered drug, to yield products of greater concentration than the maceration methods.
Large-scale Extraction Procedures
Large-scale operation demands modification of many extraction processes, where the small-scale directions are
- inappropriate. Thus, `shaking occasionally' presents no
problems with a jar containing 500 or 1000 ml of tincture. An industrial batch is likely to be 2000 litres. It is difficult to shake such vessel thus there are alternative methods of agitation. Also for economy less solvent must be needed and evaporation requirements for concentrated products are reduced.
CIRCULATORY EXTRACTION
The efficiency of extraction in a maceration process can be improved by arranging the solvent to be continuously circulated through the drug. Where solvent is pumped from the bottom of the vessel to the inlet where it is distributed through spray nozzles over the surface of the drug.
Spray nozzles Drug Product Pump
Circulatory Extraction
The movement of the solvent reduces boundary layers, and the uniform distribution minimizes local concentrations, leading to more efficient extraction in a shorter time. Like the normal maceration process, extraction is incomplete, since mass transfer will cease when an equilibrium is set up. This problem can be overcome by using a multi-stage process.
Spray nozzles Drug Product Pump
Circulatory Extraction
MULTIPLE STAGE EXTRACTION
The equipment needed for this method is a vessel for the drug, together with a circulating pump and spray distributors, and a number of tanks to receive the extracted solution. The extractor and tanks are connected with piping and valves, so that any
- ne of the tanks may be connected to
the extractor for the transfer of solution.
Principle of operation:
- 1. Fill extractor with drug, add solvent and circulate,
Run off to receiver 1.
- 2. Refill extractor with solvent and circulate.
Run off to receiver 2.
- 3. Refill extractor with solvent and circulate.
Run off to receiver 3.
Multiple Stage Extraction
- 4. Remove drug from extractor and re-charge.
Return solution from 1 to extractor. Remove for evaporation.
- 5. Return solution from 2 to extractor and circulate.
Run off to receiver 1.
Multiple Stage Extraction
- 6. Return solution from 3 to extractor and circulate. Run off
to receiver 2.
- 7. Add fresh solvent to extractor and circulate. Run off to
receiver 3.
- 8. Remove drug from extractor and re-charge. Repeat cycle.
Thus, each batch of drug is treated several times with solvent and that, once the cycle is in operation, the receivers contain solution with the strongest in receiver 1 and the weakest in receiver 3.
Multiple Stage Extraction
The drug is extracted as many times as there are
receivers (in this case three). If more extraction stages are required, it is only necessary to have more receivers.
The last treatment of the drug before it is discharged is
with fresh solvent, giving maximum extraction.
The solution is in contact with fresh drug before removal
for evaporation, giving the highest possible concentration.
Advantages
Continuous Extraction
British Pharmacopoeia method
The previous processes for the manufacture
- f
concentrated preparations have involved extraction, followed by evaporation of the solvent. Continuous extraction combines the two operations, so that, immediately after contact with the drug, the solution is evaporated and the vapour taken to a condenser while the condensed liquid is returned to the drug to continue extraction.
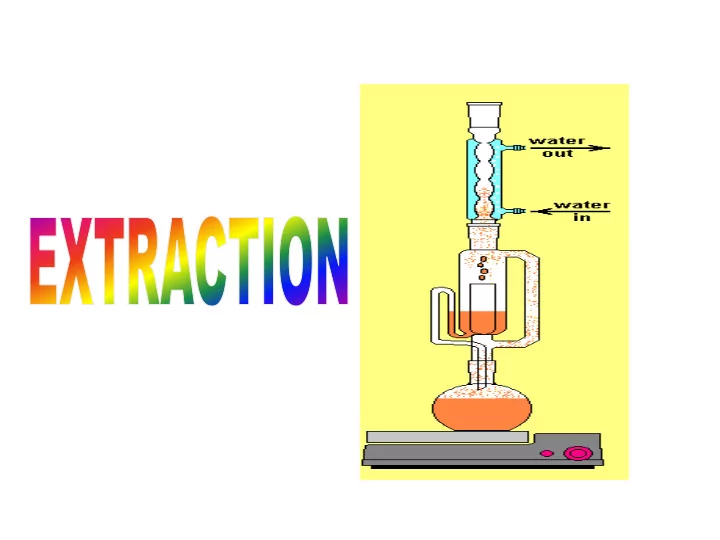
Principle
Vapour from the flask rises through the extraction chamber, Passing the drug container and condenses in the reflux condenser Then returns through the drug, taking the soluble constituents to the flask. The method is described as continuous hot extraction, since the vapour surrounds the drug container and extraction occurs at the boiling point of the solvent. Thus, extraction is a continuous percolation procedure.
Reflux
Extraction Chamber Drug Container Vapour Solution
Continuous Extraction Small-scale BP apparatus
Soxhlet apparatus
Principle
The vapour passes through the side
tube and reflux returns to the extraction chamber where the solution collects.
The liquid level will rise in the return
tube; when the liquid reaches the top of the return tube, a siphon is set up and the contents of the extraction chamber are transferred to the flask.
This method is referred to as continuous cold extraction, since the vapour is by- passed through the side arm and does not enter the extraction chamber directly. The extraction is a series of short macerations, compared with the percolation of the previous method.
Reflux
Vapour tube
Solution Return tube Continuous Extraction Small Scale Plant Soxhlet apparatus
Similar methods can be used in large-scale production; The principles of operation will be seen to resemble the laboratory equipment.
Continuous Extraction Large Scale Plant
(a) Less solvent is needed than in the conventional method, yielding concentrated products. (b) The drug is treated continuously with pure solvent, ensuring maximum concentration gradient and complete exhaustion. (c) Extraction can be continued as necessary to exhaust the
- drug. Thus, depending on the character of the drug, it is
possible to prolong extraction without increasing the volume of the product, corresponding to a drug/percolate ratio of any value up to infinity.
Advantages
(a) The drug must be powdered, so that the method is limited in a similar manner to percolation. (b) The solution is boiled continuously. This involves a considerable heat usage, as well as rendering the method unsatisfactory with thermolabile drugs. This problem can be improved by operation under vacuum.
Disadvantages
(c) The solvent is boiled continuously, so that the method is restricted to pure boiling solvents as light petroleum. This prevents the use of continuous extraction for many products where the solvent is ethanol/water system. The combined effect of these factors limit the use of the method, as in the extraction of fixed oils from seeds, with a solvent such as light petroleum.
Fa Fact ctors
- rs Af
Affec ecting ting Choi
- ice
ce of
- f
Ext xtrac raction tion Pro roce cess
Use maceration in case of: soft (such as gentian), unpowderable (such as squill), unorganised drug (such as benzoin) and when preferable to avoid powdering (such as senna fruits). Use percolation in case of hard and tough (such as nux vomica).
CHARACTER OF DRUG
If the drug has therapeutic value, use percolation for maximum extraction as in belladonna. If the drug has little therapeutic value, use maceration that the efficiency of extraction is unimportant as flavours (lemon), or bitters (gentian).
THERAPEUTIC VALUE OF THE DRUG
From the economic point of view, it is desirable to obtain complete extraction of an expensive drug (as ginger), so that percolation should be used; For cheap drugs, the reduced efficiency of maceration is acceptable due to the lower cost of the process.
STABILITY OF DRUG
Continuous extraction should be avoided when the constituents of the drug are thermolabile.
COST OF DRUG
For dilute products such as tinctures can be made by maceration or percolation, depending. For semi-concentrated preparations (concentrated infusions) the more efficient percolation process is used, unless the drug cannot be powdered double or triple maceration is chosen. For concentrated preparations ( liquid extracts or dry extracts) are made by percolation continuous extraction can be used if the solvent is suitable and the constituents are thermostable.
CONCENTRATION OF PRODUCT
it is saturated with solvent that is recovered for economy.
- In some cases the drug is pressed
as part of the extraction process, so that much of the liquid is removed.
- If
not part
- f
the process, application of pressure is a useful recovery procedure using Hydraulic Press.
Recovery of Solvent from the Marc The marc is the residue of the drug after extraction,
Hydraulic Press
- The marc, wrapped in cloth, is placed in the
perforated inner vessel, which is enclosed in another vessel having an outlet for the expressed liquid.
- Application of hydraulic pressure
to the ram presses the marc against the fixed head and expelling residual liquid.
- Recovery
- f
the remaining solvent demands vaporisation, a large area for heat transfer and an easy method of opening or
- closing. Vacuum oven can be
used for solvent recovery.
Hydraulic Press
When large quantities of drug processed, the transfer of the marc from the extraction vessel to a press and then to a recovery involves too much effort. It is preferable to recover the solvent directly from the drug while it is still in the extraction vessel. This can be done by surrounding the vessel with a steam jacket, enabling the contents to be heated when extraction has been completed. But the method is inefficient that the marc shrinks as it approaches dryness.
In the method illustrated in the figure steam is admitted to the base of the extraction vessel; heating is uniform and heat transfer is much more rapid, so that recovery of the solvent is more efficient. The distillation stage is needed for purification that vapours that pass to the condenser consist of the solvent vapour mixed with steam.
solvent recovery from extraction vessel