SLIDE 1
Evidence for resonance stabilization: enthalpies of hydrogenation - - PowerPoint PPT Presentation
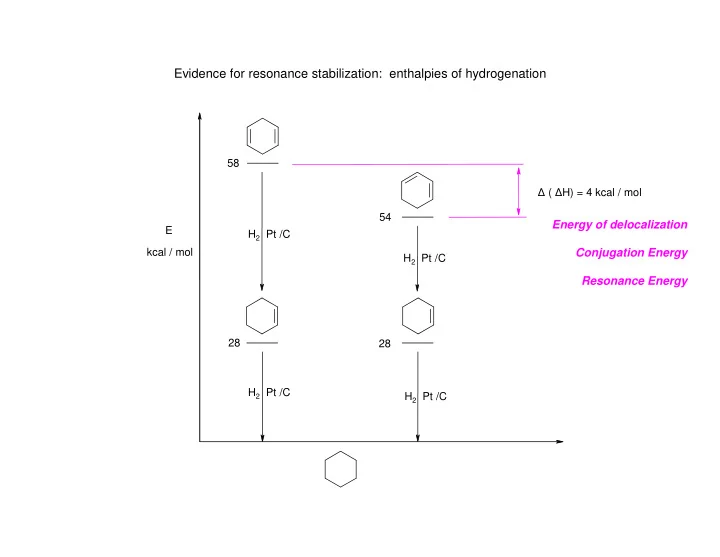
Evidence for resonance stabilization: enthalpies of hydrogenation - - PowerPoint PPT Presentation
Evidence for resonance stabilization: enthalpies of hydrogenation 58 ( H) = 4 kcal / mol 54 Energy of delocalization E H 2 Pt /C Conjugation Energy kcal / mol H 2 Pt /C Resonance Energy 28 28 H 2 Pt /C H 2 Pt /C Evidence for
SLIDE 2
SLIDE 3
Consider (E)-1,3-pentadiene:
SLIDE 4
E kcal / mol C1= C2--C3= C4 torsion, degrees 2.8 3.9 0 90 180 H H H H H H H H H H H H s - trans conformation s - cis conformation
Evidence for resonance stabilization: conformational preferences Just as there is a barrier to rotation about the C2=C3 of cis and trans-2-butene, there is a barrier to rotation about the C2 - C3 in 1,3-butadiene:
SLIDE 5
Synthesis of conjugated dienes:
- Just like alkene synthesis, only twice – conjugated dienes prefer to form as major products because they are
the most thermodynamically stable products
- Dehydrohalogenation of allylic halides
- Isolated dienes form only when it is impossible to form the conjugated isomer
Br CH3CH2O- Na+ CH3CH2OH, 90 %
SLIDE 6
What would the E1 outcome of dehydrobromination of 4-bromo-2,3,3-trimethylhexene be?
Br CH3CH2OH, ?
SLIDE 7
Reactions of conjugated dienes:
- Additions of HX
Br Br HBr dark, -80 C + 81 % 19 %
SLIDE 8
Reactions of conjugated dienes:
- Additions of HX
1,2-addition 1,4-addition Markovnikov addition product conjugate addition product
Br Br HBr dark, -80 C + 81 % 19 %
SLIDE 9
What’s the mechanism?
H Br Br Br + + Br- + + Br- +
SLIDE 10
C C H
H
C CH3
H
Br Br Br C C H
H
C CH3
H
C C H
H
C CH3
H
+ . . . . + + + + + + + + Br- Br- Br-
1 2 3 4 1 2 3 4 1 2 3 4
1,2-addition 1,3-addition 1,4-addition
Looking at the carbocation intermediate more closely: what possible addition products can form? less stable isomer never observed more stable isomer
SLIDE 11
Reactions of conjugated dienes:
- Additions of HX
1,2-addition 1,4-addition major minor 1,2-addition 1,4-addition minor major
Br Br HBr dark, -80 C + 81 % 19 % Br Br HBr dark, 45 C + 19 % 81 %
SLIDE 12
Energy vs reaction coordinate diagram:
Br Br + HBr + Br- +
SLIDE 13
Energy vs reaction coordinate diagram:
Br Br + HBr + Br- +
SLIDE 14
Energy vs reaction coordinate diagram:
Br Br + HBr + Br- +
SLIDE 15
Br Br + HBr + Br- +
Energy vs reaction coordinate diagram: For the second step, Eact < Eact so the rate of 1,2-addtion > the rate of 1,4-addition 1,2-addition 1,4-addition When the major product that forms is the fastest forming product, a reaction is said to be under rate or kinetic control.
SLIDE 16
Br Br + HBr + Br- +
Energy vs reaction coordinate diagram: For the second step, if the temperature of the reaction is Eact reverse then the 1,2- addition product can equilibrate to the the more stable 1,4-addition product 1,2-addition 1,4-addition When the major product that forms is the most stable product, a reaction is said to be under equilibrium, thermodynamic or stability control.
SLIDE 17
The Alder Rule diastereoselection is such that endo products predominate:
O OCH3 O OCH3 O OCH3 O OCH3 O OCH3 +
- r
exo endo
SLIDE 18
exo transition state: C=O carbon hoovers over CH2 of 1,3-cyclopentadiene more stable endo transition state: C=O carbon hoovers endo adduct
- ver forming C2–C3 C=C of the cyclopentadiene
SLIDE 19
C C H H H H
E C-C
- C-H
- C-C
- C-C
- *
C-H
- *
C-C
- *
(homo) (lumo)
Ultraviolet - Visible (UV-Vis) Spectroscopy Consider the ground state electronic configuration of ethene:
SLIDE 20
C C H H H H
E C-C
- C-H
- C-C
- C-C
- *
C-H
- *
C-C
- *
(homo) (lumo)
The smallest E that will promote a bonding electron to an antibonding orbital is: E = UV-Vis (with λ = 162 nm here)
SLIDE 21
λmax = 175 λmax = 245
λmax = 275 λmax = 215 Absorbance wavelength, nm
SLIDE 22
O = 280 nm A: *
SLIDE 23
O O = 204 nm A: * B: n * = 254 nm
SLIDE 24
C=O π electron transitions LUMO π* 254 nm 204 nm HOMO n HOMO-1 π
SLIDE 25