SLIDE 1
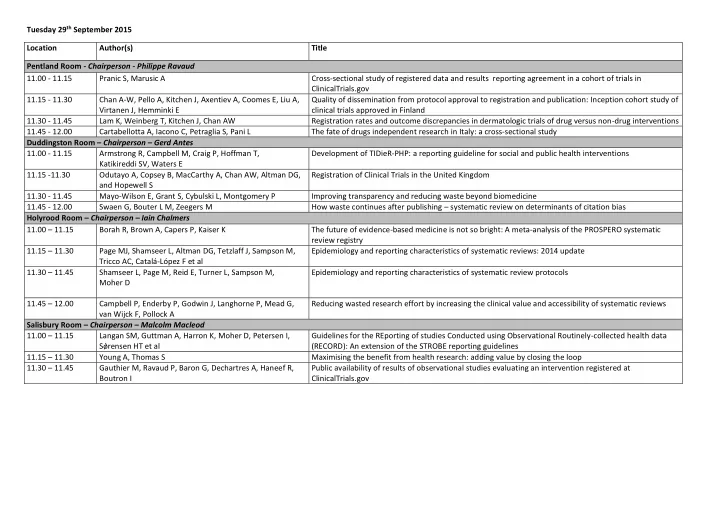
Tuesday 29th September 2015 Location Author(s) Title Pentland Room - Chairperson - Philippe Ravaud 11.00 - 11.15 Pranic S, Marusic A Cross-sectional study of registered data and results reporting agreement in a cohort of trials in ClinicalTrials.gov 11.15 - 11.30 Chan A-W, Pello A, Kitchen J, Axentiev A, Coomes E, Liu A, Virtanen J, Hemminki E Quality of dissemination from protocol approval to registration and publication: Inception cohort study of clinical trials approved in Finland 11.30 - 11.45 Lam K, Weinberg T, Kitchen J, Chan AW Registration rates and outcome discrepancies in dermatologic trials of drug versus non-drug interventions 11.45 - 12.00 Cartabellotta A, Iacono C, Petraglia S, Pani L The fate of drugs independent research in Italy: a cross-sectional study Duddingston Room – Chairperson – Gerd Antes 11.00 - 11.15 Armstrong R, Campbell M, Craig P, Hoffman T, Katikireddi SV, Waters E Development of TIDieR-PHP: a reporting guideline for social and public health interventions 11.15 -11.30 Odutayo A, Copsey B, MacCarthy A, Chan AW, Altman DG, and Hopewell S Registration of Clinical Trials in the United Kingdom 11.30 - 11.45 Mayo-Wilson E, Grant S, Cybulski L, Montgomery P Improving transparency and reducing waste beyond biomedicine 11.45 - 12.00 Swaen G, Bouter L M, Zeegers M How waste continues after publishing – systematic review on determinants of citation bias Holyrood Room – Chairperson – Iain Chalmers 11.00 – 11.15 Borah R, Brown A, Capers P, Kaiser K The future of evidence-based medicine is not so bright: A meta-analysis of the PROSPERO systematic review registry 11.15 – 11.30 Page MJ, Shamseer L, Altman DG, Tetzlaff J, Sampson M, Tricco AC, Catalá-López F et al Epidemiology and reporting characteristics of systematic reviews: 2014 update 11.30 – 11.45 Shamseer L, Page M, Reid E, Turner L, Sampson M, Moher D Epidemiology and reporting characteristics of systematic review protocols 11.45 – 12.00 Campbell P, Enderby P, Godwin J, Langhorne P, Mead G, van Wijck F, Pollock A Reducing wasted research effort by increasing the clinical value and accessibility of systematic reviews Salisbury Room – Chairperson – Malcolm Macleod 11.00 – 11.15 Langan SM, Guttman A, Harron K, Moher D, Petersen I, Sǿrensen HT et al Guidelines for the REporting of studies Conducted using Observational Routinely-collected health data (RECORD): An extension of the STROBE reporting guidelines 11.15 – 11.30 Young A, Thomas S Maximising the benefit from health research: adding value by closing the loop 11.30 – 11.45 Gauthier M, Ravaud P, Baron G, Dechartres A, Haneef R, Boutron I Public availability of results of observational studies evaluating an intervention registered at ClinicalTrials.gov

SLIDE 2
Tuesday 29th September 2015 Location Author(s) Title Pentland Room– Chairperson – John Ioannidis 14.00 – 14.15 MacLeod MR on behalf of the IICARus and NPQIP investigators Strategies for publishers to improve the reporting of in vivo research 14.15 – 14.30 Leenaars M, Ritskes-Hoitinga M Lessons learned from the implementation of systematic reviews in laboratory animal science 14.30 – 14.45 Holman C, Diamantaras A, Grittner U,Kimmelman J, Piper SK, Siegerink B, Dirnagl U Where Have All the Rodents Gone? The Effects of Attrition in Experimental Research on Cancer and Stroke 14.45 – 15.00 Danborg PB, Simonsen AL, Hrǿbjartsson A, Gǿtzsche PC Risk of bias assessment in preclinical research supporting clinical practice 15.00 – 15.15 Sena ES, on behalf of the MultiPART consortium Multi centre animal studies: a strategy to improve the prospects of translational success 15.15 – 15.30 Offenhauser N, Kurreck C, Dirnagl U, for the Department of Experimental Neurology QM Steering Group From policies to standards: Implementing and managing a structured ISO 9001 certified Quality System in an academic translational neuroscience research lab Duddingston Room – Chairperson – Ana Marusic 14.00 – 14.15 Galipeau J, Cobey KD, Shamseer L, Moher D The Role of Publication Officers in Increasing Value and Decreasing Waste in Biomedical Research 14.15 – 14.30 Shamseer L, Barbour V, Bell-Syer S, Cumpston M, Deeks J, Garner P, MacLehose H, Straus S et al Developing Core Competencies for Scientific Editors of Biomedical Journals 14.30 – 14.45 Callender T Reporting Carbon to Reduce Waste and Improve Efficiency – The GREENER Initiative 14.45 – 15.00 Toews I, Wolff RF, Binder N, Toprak G, von Elm E, Meerpohl JJ Editorial policies of hematology and oncology journals: A cross-sectional study of author instructions 15.00 – 15.15 The Transparency and Openness Promotion (TOP) Committee The TOP Guideline to promote transparency, openness, and reproducibility 15.15 – 15.30 Rodríguez AC Making the case for creating synergy between EQUATOR and manuscript editing companies Holyrood Room – Chairperson – Doug Altman 14.00 – 14.15 Kapadia M, Moher D, Offringa M, on BEHALF OF PRISMA-
PC and PRISMA-C STEERING GROUP Increasing value and reducing waste in child health systematic reviews and meta-analyses - Protocols (PRISMA-PC) and Reporting (PRISMA-C)
14.15 – 14.30 Clyburne-Sherin A, Thurairajah P, Chan WWY, Kapadia M,
Moher D, Klassen T, Offringa M SPIRIT-C: Increasing research value / reducing research waste through evidence-based reporting standards for pediatric clinical trial protocols
14.30 – 14.45 Clyburne-Sherin A, Thurairajah P, Chan WWY, Kapadia M,
Moher D, Chan A, Klassen T, Offringa M CONSORT-C: Increasing research value / reducing research waste through evidence-based reporting standards for pediatric clinical trials
14.45 – 15.00 Kapadia M, Thurairajah P, Terwee C, Beaton D, Offringa
M New evidence and consensus based reporting Instrument for the Selection of Pediatric Endpoints in Clinical Trials: InSPECT
15.00 – 15.15 Kelly LE, Offringa M
Increasing value and reducing waste in child health research through evidence-based guidance for selecting a comparator treatment in pediatric clinical trials
15.15 – 15.30 Kapadia M, Kelly LE, Needham A, Chan W, Clyburne-
Sherin A, Thurairajah P, Offringa M Increasing value and reducing waste in child health research by developing and implementing novel methodology: Enhancing Research Impact in Child Health (EnRICH)
Salisbury Room – Chairperson – Gerd Antes 14.00 – 14.15 Barnett A, Clarke P, Herbert D Randomness and high cost for funding outcomes 14.15 – 14.30 Shamseer L, Moher D, Maduekwe O, Turner L, Barbour V, Burch R et al Comparison of characteristics potential predatory journals with subscription and open access journals. A cross-sectional study. 14.30 – 14.45 Olivieri J, Manfredi L, Postacchini L, Tedesco S, Leoni P, Gabriella A et al Poor adherence to methodological recommendations supports biased research results in steroid-refractory chronic Graft-versus-Host Disease: a systematic review and meta-analysis 14.45 – 15.00 Malicki M, Utrobicic A, Marusic A Addressing duplicate publications in biomedicine: a mixed method study 15.00 – 15.15 Hemminki E Improper research regulation is a cause for waste in clinical research 15.15 – 15.30 Bouter LM, Axelsen N, ter Riet G, Tijdink J Survey among experts to rank major and minor research misbehaviors

SLIDE 3 Wednesday 30th September 2015 Location Author(s) Title Pentland Room - Chairperson – Malcolm Macleod 09.30 – 09.45 Mayo-Wilson E, Fusco N, Ehmsen J, Gresham G, Heyward J, Huftless S et al Wasted results: Multiplicity in outcome definitions is a mechanism for reporting bias (oral presentation) 09.45 – 10.00 Odutayo A, Jairath V, Chan AW, Hopewell S, Altman DG Assessing the Use of Patient Reported Outcomes in Protocols for Randomized Controlled Trials Approved in the United Kingdom in 2012 10.00 - 10.15 Agha RA, Fowler AJ, Sagou H, Pidgeon TE, Wellstead G, Orgill DP THE NEED FOR CORE OUTCOME REPORTING IN AUTOLOGOUS FAT GRAFTING FOR BREAST RECONSTRUCTION 10.15 – 10.30 Dickersin K, Saldanha I, Le J, Law A, Scherer R, Li T Use of a well-known surrogate outcome instead of a patient-important outcome can be viewed as research waste: Examination of an ad hoc sample of clinical trials and systematic reviews Duddingston Room – Chairperson – An-Wen Chan 09.30 – 09.45 Boutron I, Dechartres A, Baron G, Li J, Ravaud P Increasing research value through data sharing by pharmaceutical companies 09.45 – 10.00 Sach T Could value of information methods be useful in prioritising research to address the treatment uncertainties identified by James Lind Alliance Priority setting partnerships? 10.00 - 10.15 Papathanasiou A A novel type of literature review that promotes more useful research 10.15 – 10.30 Job DE, Danso S, Dickie DA, Rodriguez D, Robson A, Pernet C, Shenkin SD, Wardlaw JM Reuse of brain imaging and clinical data from completed studies across the life course: the Brain Images
- f Normal Subjects (BRAINS) Imagebank.
Holyrood Room – Chairperson – Gerd Antes 09.30 – 09.45 Nasser M, Glasziou P, Chalmers I What do medical research funders do to increase value and reduce waste in research? 09.45 – 10.00 Ferri M, Bo A, Oliver S, Moreira M, Amato L, Davioli M Avoiding waste in research on treatment of drug dependence: a systematic approach by the European Monitoring Centre on Drugs and Drug Dependence and the European Commission 10.00 - 10.15 Hemkens LG, Benchimol EI, Langan SM, Briel M, Kasenda B, Januel JM et al REPORTING OF STUDIES USING ROUTINELY COLLECTED HEALTH DATA: SYSTEMATIC LITERATURE ANALYSIS 10.15 – 10.30 Ndounga Diakou LA, Ntoumi F, Bafeta A, Boutron I Waste in the conduct and reporting of randomized controlled trials performed in Sub-Saharan Africa Salisbury Room – Chairperson – Paul Glasziou 09.30 – 09.45 Hermann-Lingen C, Brunner E, Hildenbrand S, Loew TH, Raupach T, Spies C et al Evaluation of medical research performance – the position of the Association of the Scientific Medical Societies in Germany (AWMF) 09.45 – 10.00 Report by the Nuffield Council on Bioethics The culture of scientific research in the UK 10.00 - 10.15 Needham AC, Junker A, Kimber S, Heslegrave R, Boydell K, Greenberg R et al Variability in application of research ethics principles in reviews of multicentre clinical trial protocols: Qualitative exploration of inter-institutional ethics reviews
Top Scoring Submissions

SLIDE 4
Wednesday 30th September 2015 Location Author(s) Title Pentland Room –– Chairperson – Doug Altman 11.00 - 11.15 Li T, Rouse B, Shi Q, Dickersin K Working from all angles 11.15 - 11.30 Crequit P, Trinquart L, Yavchitz A, Ravaud P Wasted research when systematic reviews fail to provide a complete and up-to-date evidence synthesis: the example of lung cancer 11.30 - 11.45 Strech D, Chin W, Hradetzky S, Silva D Informed development and implementation of registries in animal research. Findings from a systematic review and from key informant interviews 11.45 - 12.00 Loudon K Reducing waste through better trial design: the PRECIS-2 tool